AMUEEE SERIES Chemistry
Coordination Compounds
25 previous year questions.
Volume: 25 Ques
Yield: High
High-Yield Trend
3
2018 1
2016 6
2013 4
2012 5
2011 6
2010 Chapter Questions 25 MCQs
01
PYQ 2010
easy
chemistry ID: amueee-2
Which of the following is an outer orbital complex ?
1
2
3
4
02
PYQ 2010
medium
chemistry ID: amueee-2
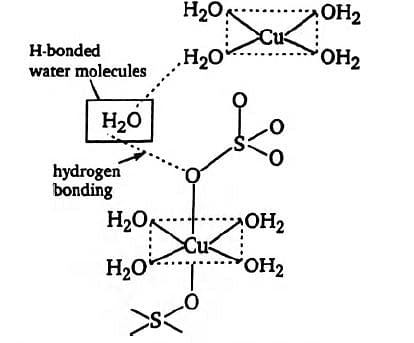
How many hydrogen bonded water molecule are associated with ?
1
2
3
4
03
PYQ 2010
easy
chemistry ID: amueee-2
Which of the following is Vaska?s compound ?
04
PYQ 2010
easy
chemistry ID: amueee-2
Which of the following configuration can undergo distortion ?
05
PYQ 2010
medium
chemistry ID: amueee-2
What is the magnetic moment of ion in ?
1
2
3
4
None of these
06
PYQ 2010
medium
chemistry ID: amueee-2
The most stable complex among the following is
1
2
3
4
07
PYQ 2011
easy
chemistry ID: amueee-2
The hybridisation states of , and species are respectively
08
PYQ 2011
medium
chemistry ID: amueee-2
Which of the following has the largest number of isomers?
1
2
3
4
09
PYQ 2011
medium
chemistry ID: amueee-2
Which of the following is an outer orbital complex?
1
2
3
4
10
PYQ 2011
medium
chemistry ID: amueee-2
Which one of the following is heteroleptic complex?
1
2
3
4
11
PYQ 2011
medium
chemistry ID: amueee-2
Weak field ligands form high spin complexes due to (where is crystal field splitting and = pairing energy)
1
2
3
4
12
PYQ 2012
medium
chemistry ID: amueee-2
Which of the following is not a - bonded complex?
1
Zeise's salt
2
Ferrocene
3
Dibenzene chromium
4
Tetramethyl lead
13
PYQ 2012
easy
chemistry ID: amueee-2
Compounds, and , shows the following type of isomerism
1
hydrate isomerism
2
ionisation isomerism
3
linkage isomerism
4
coordination position isomerism
14
PYQ 2012
medium
chemistry ID: amueee-2
Effective atomic number of the central metal ion, , in the complex is: (The atomic no. of is )
1
74
2
90
3
86
4
84
15
PYQ 2012
medium
chemistry ID: amueee-2
, a complex ion of cobalt (III), absorbs radiations in violet region of the visible light. Its aqueous solution, therefore, appears
1
pink
2
orange
3
blue
4
yellow
16
PYQ 2013
medium
chemistry ID: amueee-2
What is the oxidation state of in
1
2
2
3
3
1
4
4
17
PYQ 2013
medium
chemistry ID: amueee-2
The structure of paramagnetic nickel complex, is
1
tetrahedral
2
square planar
3
trigonal bipyramidal
4
distorted octahedral
18
PYQ 2013
medium
chemistry ID: amueee-2
Wilkinson's catalyst is used for
1
epoxidation
2
hydrogenation
3
polymerisation
4
substitution
19
PYQ 2013
medium
chemistry ID: amueee-2
Predict the correct shape of .
1
Pyramidal,
2
Octahedral,
3
Tetrahedral,
4
None of these
20
PYQ 2013
medium
chemistry ID: amueee-2
and are examples of which type of isomerism ?
1
Linkage
2
Geometrical
3
Ionisation
4
Optical
21
PYQ 2013
medium
chemistry ID: amueee-2
The unpaired electron in is
1
1
2
2
3
3
4
0
22
PYQ 2016
medium
chemistry ID: amueee-2
For complexes and which one of the following statement is true
1
is diamagnetic while is paramagnetic and both the complexes have square planar geometry
2
is paramagnetic while is diamagnetic and both the complexes have tetrahedral geometry
3
is paramagnetic while is diamagnetic and both the complexes have square planar geometry
4
is diamagnetic while is paramagnetic and both the complexes have tetrahedral geometry
23
PYQ 2018
easy
chemistry ID: amueee-2
The value is
1
2
3
4
24
PYQ 2018
medium
chemistry ID: amueee-2
Which of the following complexes is optically active?
1
2
3
4
25
PYQ 2018
medium
chemistry ID: amueee-2
The crystal field splitting energy for is about What would be the value of ?
1
2
3
4





 undefined
undefined





