IIT-JAM-CY SERIES Physical-chemistry
Chemical Equilibria
9 previous year questions.
Volume: 9 Ques
Yield: Medium
High-Yield Trend
4
2024 3
2023 1
2022 1
2018 Chapter Questions 9 MCQs
01
PYQ 2018
medium
physical-chemistry ID: iit-jam-
The solubility of in 0.10 M KI(aq) is ........... M (rounded up to two decimal places).
]}\)
02
PYQ 2022
hard
physical-chemistry ID: iit-jam-
The pH of an aqueous buffer prepared using CH3COOH and CH3COO Na+ is 4.80.
The quantity is _____. (round off to three decimal places)
[Given: pKa of CH3COOH in water is 4.75]
The quantity is _____. (round off to three decimal places)
[Given: pKa of CH3COOH in water is 4.75]
03
PYQ 2023
medium
physical-chemistry ID: iit-jam-
The isoelectric point of glutamic acid is ______.
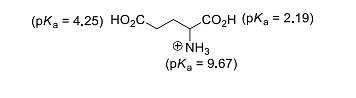
(round off to two decimal places)

(round off to two decimal places)
04
PYQ 2023
medium
physical-chemistry ID: iit-jam-
The volume of water (in mL) required to be added to a 100 mL solution (aq. 0.1 M) of a weak acid (HA) at 25 °C to double its degree of dissociation is
[Given: Ka of HA at 25 °C=1.8 x10−5 ]
[Given: Ka of HA at 25 °C=1.8 x10−5 ]
1
100
2
200
3
300
4
400
05
PYQ 2023
easy
physical-chemistry ID: iit-jam-
The following diagram is obtained in a pH-metric titration of a weak dibasic acid (H2A) with a strong base. The point that best represents [HA−]=[A −2 ] is
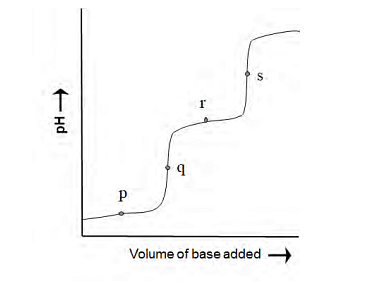

06
PYQ 2024
medium
physical-chemistry ID: iit-jam-
0.1 M aqueous solution of a weak monobasic acid has pH 2.0. The pKa of the monobasic acid is _______. (rounded off to one decimal place)
07
PYQ 2024
medium
physical-chemistry ID: iit-jam-
Consider the exothermic chemical reaction O2(𝑔)+2H2(𝑔) ⇌ 2H2O(𝑔) at equilibrium in a closed container. The correct statement(s) is/are
1
At equilibrium, introduction of catalyst increases product formation
2
Equilibrium constant decreases with increase in temperature.
3
The equilibrium constant 𝐾𝑃 increases with pressure.
4
Decrease in volume of reaction vessel increases product formation.
08
PYQ 2024
easy
physical-chemistry ID: iit-jam-
The ratio of osmotic pressures of aqueous solutions of 0.01 M BaCl2 to 0.005 M NaCl is
[Given: Both compounds dissociate completely in water]
[Given: Both compounds dissociate completely in water]
1
3:1
2
1:4
3
1:1
4
3:2
09
PYQ 2024
medium
physical-chemistry ID: iit-jam-
A 1.0 L solution is prepared by dissolving 2.0 g of benzoic acid and 4.0 g of sodium benzoate in water. The pH of the resulting solution is _______. (rounded off to one decimal place)
Given: Molar mass of benzoic acid is 122 g mol−1
Molar mass of sodium benzoate is 144 g mol−1
p𝐾a of benzoic acid is 4.2
Given: Molar mass of benzoic acid is 122 g mol−1
Molar mass of sodium benzoate is 144 g mol−1
p𝐾a of benzoic acid is 4.2