IIT-JAM-CY SERIES Physical-chemistry
Chemical Kinetics
15 previous year questions.
Volume: 15 Ques
Yield: Medium
High-Yield Trend
4
2024 4
2023 1
2022 3
2020 3
2018 Chapter Questions 15 MCQs
01
PYQ 2018
medium
physical-chemistry ID: iit-jam-
The reaction, Products, follows first-order kinetics. If represents the concentration of reactant at time , the INCORRECT variation is shown in 
1
(A)
2
(B)
3
(C)
4
(D)
02
PYQ 2018
medium
physical-chemistry ID: iit-jam-
The time for 50% completion of a zero-order reaction is 30 min. Time for 80% completion of this reaction is ......... min.
03
PYQ 2018
medium
physical-chemistry ID: iit-jam-
decays to one-fourth of its initial amount in 29.8 hours. Its decay constant is ........... hour (rounded up to four decimal places).
04
PYQ 2020
medium
physical-chemistry ID: iit-jam-
A bacterial colony grows via cell division where each mother bacterium independently produces two daughter cells in 20 minutes. If the concentration of bacteria is , the colony becomes harmful. Starting from a colony with an initial concentration of 5 cm , the time taken (in minutes) for the colony to become harmful is ......... (Round off to nearest integer)
05
PYQ 2020
medium
physical-chemistry ID: iit-jam-
For the reaction shown in Scheme 1, the concentration profiles of different species are provided.

Based on this graph, the correct condition(s) regarding the rate constants is(are)
1
2
3
4
06
PYQ 2020
medium
physical-chemistry ID: iit-jam-
For the radical chain reaction below, the correct classification for step 2 and step 3 is, respectively,
Step 1:
Step 2:
Step 3:
Step 1:
Step 2:
Step 3:
1
chain propagating, chain terminating
2
chain branching, chain terminating
3
chain propagating, chain propagating
4
chain propagating, chain branching
07
PYQ 2022
medium
physical-chemistry ID: iit-jam-
For the reaction
the rate expression is . If the concentration of is
the rate expression is . If the concentration of is
1
tripled
2
halved
3
doubled
4
unchanged
08
PYQ 2023
easy
physical-chemistry ID: iit-jam-
For the elementary reaction  At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of is ______. (round off to the nearest integer)
At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of is ______. (round off to the nearest integer)
 At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of is ______. (round off to the nearest integer)
At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of is ______. (round off to the nearest integer) 09
PYQ 2023
easy
physical-chemistry ID: iit-jam-
An elementary reaction 2A→P follows a second order rate law with rate constant 2.5×10−3 dm3 mol−1 s−1 . The time required for the concentration of A to change from 0.4 mol dm−3 to 0.2 mol dm−3 is ___s.
(round off to the nearest integer)
(round off to the nearest integer)
10
PYQ 2023
easy
physical-chemistry ID: iit-jam-
For the given elementary reactions, the steady-state concentration of X is


11
PYQ 2023
easy
physical-chemistry ID: iit-jam-
The amount of ethane produced in the following reaction is kg.

(round off to two decimal places)

(round off to two decimal places)
12
PYQ 2024
medium
physical-chemistry ID: iit-jam-
For a zero-order reaction P→Q, the concentration of P becomes half of its initial concentration in 30 minutes after starting the reaction.
The concentration of P becomes zero at ____ minutes. (rounded off to the nearest integer)
The concentration of P becomes zero at ____ minutes. (rounded off to the nearest integer)
13
PYQ 2024
easy
physical-chemistry ID: iit-jam-
The function exp(−2(𝑥-1)2) attains a maximum at 𝑥= _______. (rounded off to the nearest integer)
14
PYQ 2024
medium
physical-chemistry ID: iit-jam-
In the oxidation of phosphorus with oxygen, 0.2 mol of P4 produces ____ g of P4O10.
(rounded off to one decimal place)
[Given: Atomic weight of P = 31; Atomic weight of O = 16]
(rounded off to one decimal place)
[Given: Atomic weight of P = 31; Atomic weight of O = 16]
15
PYQ 2024
medium
physical-chemistry ID: iit-jam-
For the reaction
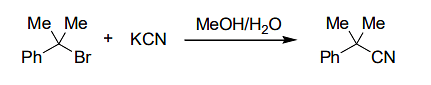
if the concentration of KCN is increased four times, then the rate of the reaction would be
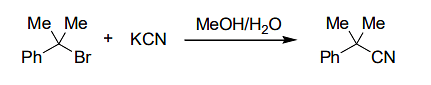
if the concentration of KCN is increased four times, then the rate of the reaction would be
1
unaffected
2
increased by two times.
3
decreased by four times.
4
increased by four times.