IIT-JAM-CY SERIES Physical-chemistry
Thermodynamics
23 previous year questions.
Volume: 23 Ques
Yield: High
High-Yield Trend
3
2024 4
2023 7
2022 4
2020 5
2018 Chapter Questions 23 MCQs
01
PYQ 2018
medium
physical-chemistry ID: iit-jam-
The CORRECT expression that corresponds to reversible and adiabatic expansion of an ideal gas is
1
2
3
4
02
PYQ 2018
easy
physical-chemistry ID: iit-jam-
The CORRECT expression(s) for isothermal expansion of 1 mol of an ideal gas is(are)
03
PYQ 2018
medium
physical-chemistry ID: iit-jam-
Consider the reaction .
The value of for the reaction at 300 K is -281.8 kJ mol . The value of at same temperature is ......... kJ mol .
[R = 8.3 J K mol ]
04
PYQ 2018
medium
physical-chemistry ID: iit-jam-
The value of for 1 mol of gas predicted from the principle of equipartition of energy, ignoring vibrational contribution, is .......... J K mol (rounded up to two decimal places).
05
PYQ 2018
medium
physical-chemistry ID: iit-jam-
For the reaction , the following information is given: The value of for the reaction is ........... J K mol .
06
PYQ 2020
medium
physical-chemistry ID: iit-jam-
The heat of formation of MgO at 300 K and 1 bar pressure is -600.60 kJ mol . The free energy (in kJ mol ) of formation of MgO at 280 K is ......... (Round off to nearest integer)
07
PYQ 2020
medium
physical-chemistry ID: iit-jam-
Sea water containing 1 M NaCl has to be desalinated at 300 K using a membrane permeable only to water. The minimum pressure (in bars) required on the sea-water side of the membrane is ......... (Round off to one decimal place)
08
PYQ 2020
medium
physical-chemistry ID: iit-jam-
In water, the enthalpy of a protein in its folded state ( ) is lower than that in its unfolded state ( ). The entropies of the folded and unfolded states are and , respectively. The condition(s) under which this protein spontaneously folds in water at a temperature , is(are)
1
2
3
4
09
PYQ 2020
easy
physical-chemistry ID: iit-jam-
The graph that represents the temperature (T) – entropy (S) variation of a Carnot cycle is

10
PYQ 2022
medium
physical-chemistry ID: iit-jam-
The standard Gibbs free energy change for the reaction
at 2500 K is
The equilibrium constant for the reaction is
[Given: ]
at 2500 K is
The equilibrium constant for the reaction is
[Given: ]
1
0.994
2
1.006
3
3.42 x
4
292.12
11
PYQ 2022
hard
physical-chemistry ID: iit-jam-
Among the following, the reaction(s) that favor(s) the formation of the products at 25 °C is/are
1

2

3

4

12
PYQ 2022
medium
physical-chemistry ID: iit-jam-
The reaction involve(s)


1
migratory insertion
2
change in electron count of Rh from 18 to 16
3
oxidative addition
4
change in electron count of Rh from 16 to 18
13
PYQ 2022
medium
physical-chemistry ID: iit-jam-
For an N-atom nonlinear polyatomic gas, the constant volume molar heat capacity Cv,m has the expected value of 3(N-1)R, based on the principle of equipartition of energy. The correct statement(s) about the measured value of Cv,m is/are
1
The measured is independent of temperature.
2
The measured is dependent on temperature.
3
The measured is typically lower than the expected value.
4
The measured is typically higher than the expected value.
14
PYQ 2022
medium
physical-chemistry ID: iit-jam-
The conversion of to involve(s)
1
the removal of an electron from a * molecular orbital of
2
an increase in the bond order from 1 in to 1.5 in
3
the formation of a paramagnetic species
4
the removal of an electron from a molecular orbital localized predominantly on Cl
15
PYQ 2022
medium
physical-chemistry ID: iit-jam-
Iron is extracted from its ore via the reaction
The volume of CO (at STP) required to produce 1 kg of iron is ________liters. (round off to the nearest integer)
[Given: Atomic wt. of Fe = 56; assume STP to be 0 °C and 1 atm]
The volume of CO (at STP) required to produce 1 kg of iron is ________liters. (round off to the nearest integer)
[Given: Atomic wt. of Fe = 56; assume STP to be 0 °C and 1 atm]
16
PYQ 2022
hard
physical-chemistry ID: iit-jam-
At constant volume, 1.0 kJ of heat is transferred to 2 moles of an ideal gas at 1 atm and 298 K. The final temperature of the ideal gas is ____K. (round off to one decimal place)
[Given: R = 8.314 J K-1 mol-1]
[Given: R = 8.314 J K-1 mol-1]
17
PYQ 2023
hard
physical-chemistry ID: iit-jam-
Consider the following reaction:
2 C6H6+15 O2→12 CO2+6 H2O =−3120 kJ mol−1.
A closed system initially contains 5 moles of benzene and 25 moles of oxygen under standard conditions at 298 K. The reaction was stopped when 17.5 moles of oxygen is left. The amount of heat evolved during the reaction is____kJ.
(round off to the nearest integer)
2 C6H6+15 O2→12 CO2+6 H2O =−3120 kJ mol−1.
A closed system initially contains 5 moles of benzene and 25 moles of oxygen under standard conditions at 298 K. The reaction was stopped when 17.5 moles of oxygen is left. The amount of heat evolved during the reaction is____kJ.
(round off to the nearest integer)
18
PYQ 2023
hard
physical-chemistry ID: iit-jam-
In the Born-Haber cycle, the heat of formation of CuCl is ______ kJ/mol.
[Given: Heat of atomization of Cu = +338 kJ/mol,
Ionization energy of Cu = +746 kJ/mol,
Heat of atomization of Cl2 = +121 kJ/mol,
Electron affinity of Cl = −349 kJ/mol, and
Lattice energy of CuCl = −973 kJ/mol]
(round off to the nearest integer)
[Given: Heat of atomization of Cu = +338 kJ/mol,
Ionization energy of Cu = +746 kJ/mol,
Heat of atomization of Cl2 = +121 kJ/mol,
Electron affinity of Cl = −349 kJ/mol, and
Lattice energy of CuCl = −973 kJ/mol]
(round off to the nearest integer)
19
PYQ 2023
medium
physical-chemistry ID: iit-jam-
The volume of 2.0 mol of an ideal gas is reduced to half isothermally at 300 K in a closed system. The value of ΔG is_____ kJ.
[Given: R=8.314 J mol-1 K-1 ]
(round off to two decimal places)
[Given: R=8.314 J mol-1 K-1 ]
(round off to two decimal places)
20
PYQ 2023
medium
physical-chemistry ID: iit-jam-
The correct relation(s) for an ideal gas in a closed system is/are
1
2
3
4
21
PYQ 2024
medium
physical-chemistry ID: iit-jam-
The enthalpy change for the reaction
C(𝑔) + O2(𝑔)→CO(𝑔) is _______ kJ per mole of CO(𝑔) produced.
(rounded off to one decimal place)
[Given:
C(𝑔)+O2(𝑔) → CO2(𝑔), ΔHrxn = −393.5 kJ per mole of CO2(𝑔) produced
CO2(𝑔) → CO(𝑔) + O2(𝑔), ΔHrxn = 283.0 kJ per mole of CO(𝑔) produced]
C(𝑔) + O2(𝑔)→CO(𝑔) is _______ kJ per mole of CO(𝑔) produced.
(rounded off to one decimal place)
[Given:
C(𝑔)+O2(𝑔) → CO2(𝑔), ΔHrxn = −393.5 kJ per mole of CO2(𝑔) produced
CO2(𝑔) → CO(𝑔) + O2(𝑔), ΔHrxn = 283.0 kJ per mole of CO(𝑔) produced]
22
PYQ 2024
hard
physical-chemistry ID: iit-jam-
A system undergoes one clockwise cycle from point X back to point X as shown in the figure below:
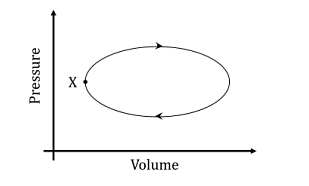
The correct statement about this process is

The correct statement about this process is
1
Internal energy of the system decreases at the end of the cycle
2
Entropy of the system increases at the end of the cycle
3
System performs work on the surroundings during the cycle.
4
Heat exchanged between system and surroundings is zero during the cycle.
23
PYQ 2024
easy
physical-chemistry ID: iit-jam-
Identify the reaction for which, at equilibrium, a change in the volume of the closed reaction vessel at a constant temperature will not affect the extent of the reaction
1
CaCO3 (𝑠) ⇌ CaO(𝑠) + CO2(𝑔)
2
H2 (𝑔) + I2 (𝑔) ⇌ 2HI(𝑔)
3
2NO2 (𝑔) ⇌ N2O4 (𝑔)
4
CO2 (𝑠) ⇌ CO2 (𝑔)