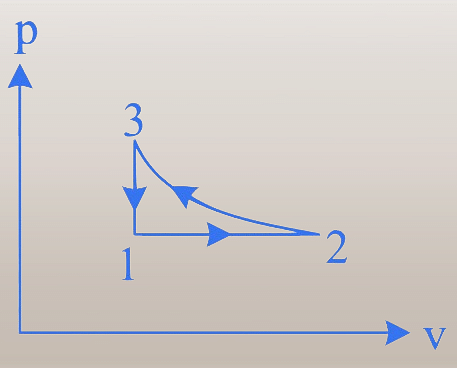
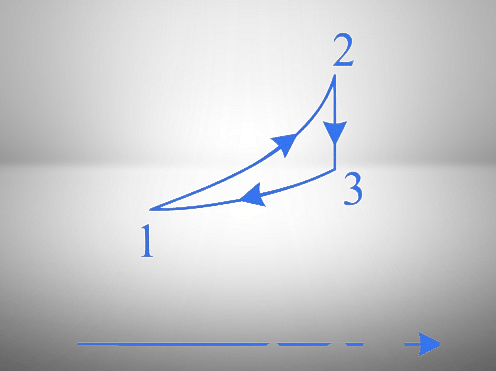
To solve the given problem, we need to analyze the cyclic process shown in both the - and - diagrams.
Analyzing the Diagrams
- Diagram:
1. Process 1 → 2: The curve shows a decrease in pressure and an increase in volume.
2. Process 2 → 3: The curve shows a decrease in both pressure and volume.
3. Process 3 → 1: The curve shows an increase in pressure with a constant volume, indicating an isochoric process.
- Diagram:
1. Process 1 → 2: The curve shows an increase in both temperature and entropy.
2. Process 2 → 3: The curve shows a decrease in temperature with constant entropy, indicating an adiabatic process.
3. Process 3 → 1: The curve shows a decrease in entropy and temperature.
Validating the Statements
Statement (B):
3 → 1: Isochoric, 2 → 3: Adiabatic
- 3 → 1: In the - diagram, volume is constant while pressure changes, confirming an isochoric process.
- 2 → 3: In the - diagram, entropy is constant while temperature changes, confirming an adiabatic process.
Thus, Statement (B) is correct.
Statement (C):
Work done by the system in the complete cyclic process is non-zero
- In a cyclic process, the work done by the system is represented by the area enclosed by the cycle in the - diagram. Since the process forms a closed loop, the enclosed area indicates non-zero work done.
Thus, Statement (C) is correct.
Statement (D):
The heat absorbed by the system in the complete cyclic process is non-zero
- According to the first law of thermodynamics for a cyclic process, the net change in internal energy over one complete cycle is zero ( ). This implies that the heat absorbed ( ) is equal to the work done ( ). Since the work done is non-zero (as confirmed in Statement (C)), the heat absorbed must also be non-zero.
Thus, Statement (D) is correct.
Conclusion:
The correct statements are:
(B) 3 → 1: Isochoric, 2 → 3: Adiabatic
(C) Work done by the system in the complete cyclic process is non-zero
(D) The heat absorbed by the system in the complete cyclic process is non-zero