JKCET SERIES Chemistry
Chemical Bonding And Molecular Structure
25 previous year questions.
Volume: 25 Ques
Yield: High
High-Yield Trend
1
2024 1
2019 1
2018 2
2017 1
2016 1
2015 3
2014 4
2013 4
2011 1
2009 3
2008 3
2007 Chapter Questions 25 MCQs
01
PYQ 2007
medium
chemistry ID: jkcet-20
The geometrical shape of hybridisation is
1
linear
2
trigonal bipyramid
3
square planar
4
tetrahedral
02
PYQ 2007
medium
chemistry ID: jkcet-20
Which one of the following examples exhibit transient existence?
1
2
3
4
03
PYQ 2007
medium
chemistry ID: jkcet-20
The number of covalent bonds in fluorine molecule is
1
2
2
3
3
1
4
5
04
PYQ 2008
medium
chemistry ID: jkcet-20
According to theory the repulsion between different pair (lone or bond) of electrons obey the order
1
lp -bp -lp -lp >bp -bp
2
lp -bp > bp -bp >lp-lp
3
lp -lp >lp -bp >lp -bp
4
bp -bp >lp -lp >lp-bp
05
PYQ 2008
medium
chemistry ID: jkcet-20
From the molecular orbital theory, one can show that the bond order in molecule as
1
2
2
1
3
3
4
4
06
PYQ 2008
medium
chemistry ID: jkcet-20
The nature of the bond in diamond is
1
ionic
2
covalent
3
metallic
4
coordinate covalent
07
PYQ 2009
medium
chemistry ID: jkcet-20
Which one of the following molecules is paramagnetic?
1
2
3
4
08
PYQ 2011
medium
chemistry ID: jkcet-20
The bond order of molecule is
1
1
2
2
3
0
4
3
09
PYQ 2011
medium
chemistry ID: jkcet-20
In which of the following molecules the central atom has two lone pairs of electrons?
1
2
3
4
10
PYQ 2011
medium
chemistry ID: jkcet-20
The hybridization involved in is
1
2
3
4
11
PYQ 2011
medium
chemistry ID: jkcet-20
Among the following which one is a linear molecule having zero dipole moment?
1
2
3
4
12
PYQ 2013
medium
chemistry ID: jkcet-20
Which of the following species has a trigonal planar shape?
1

2
3
4
13
PYQ 2013
medium
chemistry ID: jkcet-20
Select the molecule which has only one bond
1
2
3
4
14
PYQ 2013
medium
chemistry ID: jkcet-20
The bond order of on the basis of molecular orbital theory is [Atomic number of ]
1
3
2
2.5
3
2
4
1.5
15
PYQ 2013
medium
chemistry ID: jkcet-20
Which of the following molecules has the maximum dipole moment?
1
2
3
4
16
PYQ 2014
medium
chemistry ID: jkcet-20
In the formation of bond, the atomic orbitals overlap in such a way that
1
their axes remain parallel to each other and perpendicular to the internuclear axis
2
their axes remain parallel to each other and parallel to the internuclear axis
3
their axes remain perpendicular to each other and parallel to the internuclear axis
4
their axes remain perpendicular to each other and perpendicular to the internuclear axis
17
PYQ 2014
medium
chemistry ID: jkcet-20
What kinds of bonds are present in ethene molecule?
1
1 pi and 5 sigma bonds
2
1 pi and 3 sigma bonds
3
1 sigma and 1 pi bonds
4
3 sigma and 3 pi bonds
18
PYQ 2014
medium
chemistry ID: jkcet-20
Among the following molecule which has the zero dipole moment?
1
2
3
4
19
PYQ 2015
easy
chemistry ID: jkcet-20
Shape of is
1
tetrahedral
2
square planar
3
trigonal pyramid
4
see-saw
20
PYQ 2016
easy
chemistry ID: jkcet-20
The sulphur compound in which the sulphur atom has octet configuration in its valence shell among the following is
1
sulphur trioxide
2
sulphur hexafluoride
3
sulphur dichloride
4
sulphur dioxide
21
PYQ 2017
medium
chemistry ID: jkcet-20
What is the correct order of bond strength for and ?
1
2
3
4
22
PYQ 2017
medium
chemistry ID: jkcet-20
The geometry and type of hybrid orbital present around the central atom in is
1
linear,
2
trigonal planar,
3
tetrahedral,
4
pyramidal,
23
PYQ 2018
easy
chemistry ID: jkcet-20
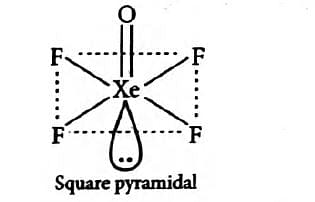
What is the hybridization and geometry of the compound ?
1
and octahedral
2
and square pyramidal
3
and trigonal bipyramidal
4
and square pyramidal
24
PYQ 2019
medium
chemistry ID: jkcet-20
is a polar solvent while is a non-polar solvent because has
1
linear structure
2
symmetrical structure
3
unsymmetrical structure
4
square planar structure
25
PYQ 2024
medium
chemistry ID: jkcet-20
Which of the following molecule doesn't involve covalent bond?
1
H O
2
CCl
3
NaCl
4
O