NEET Physics
Ideal Gas Law
Low Priority
Consistency: 20%
Weightage: 4 / 20 Yrs
High-Yield Trend
2
2025 1
2024 1
2022 2
2021 Questions 6 MCQs
01
PYQ 2021
easy
physics ID: neet-202
Choose the correct option for graphical representation of Boyle's law, which shows a graph of pressure vs. volume of a gas at different temperatures:
| 1. | |
| 2. | |
| 3. | |
| 4. |
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2021
easy
physics ID: neet-202
The correct option for the total pressure (in atm) in a mixture of 4 g O2 and 2 g H2 confined in a total volume of one liter at 0C is :
[Given R=0.082 L atm mol-1 K-1, T=273 K]
1. 25.18
2. 26.02
3. 2.518
3. 2.602
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2022
easy
physics ID: neet-202
A 10.0 L flask contains 64 g of oxygen at 27°C. (Assume gas is behaving ideally). The pressure inside the flask in the bar is:
(Given R = 0.0831 L bar K-1 mol-1)
1. 4.9
2. 2.5
3. 498.6
4. 49.8
(Given R = 0.0831 L bar K-1 mol-1)
1. 4.9
2. 2.5
3. 498.6
4. 49.8
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2024
easy
physics ID: neet-202
The following graph represents the curves of an ideal gas (where is the temperature and the volume) at three pressures and compared with those of Charles's law represented as dotted lines.
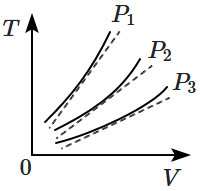
Then the correct relation is:

Then the correct relation is:
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
05
PYQ 2025
medium
physics ID: neet-202
A container has two chambers of volumes and separated by a partition made of a thermal insulator. The chambers contains and moles of ideal gas at pressures and respectively. When the partition is removed, the mixture attains an equilibrium pressure of:
1.
2.
3.
4.
1.
2.
3.
4.
1
(1)
2
(2)
3
(3)
4
(4)
06
PYQ 2025
medium
physics ID: neet-202
An oxygen cylinder of volume litre has moles of oxygen. After some oxygen is withdrawn from the cylinder, its gauge pressure drops to atmospheric pressure at temperature The mass of the oxygen withdrawn from the cylinder is nearly equal to:
Given, and molecular mass of atm pressure
Given, and molecular mass of atm pressure
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)