NEET Physics
Molar Specific Heat
Low Priority
Consistency: 20%
Weightage: 4 / 20 Yrs
High-Yield Trend
1
2018 1
2016 1
2013 1
2006 Questions 4 MCQs
01
PYQ 2006
easy
physics ID: neet-200
The molar specific heat at a constant pressure of an ideal gas is The ratio of specific heat at constant pressure to that at constant volume is:
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2013
easy
physics ID: neet-201
The molar specific heats of an ideal gas at constant pressure and volume are denoted by and respectively. If and is the universal gas constant, then is equal to:
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2016
medium
physics ID: neet-201
One mole of an ideal monatomic gas undergoes a process described by the equation The heat capacity of the gas during this process is:
| 1. | 2. | ||
| 3. | 4. | |
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2018
easy
physics ID: neet-201
The volume of a monatomic gas varies with its temperature as shown in the graph. The ratio of work done by the gas to the heat absorbed by it when it undergoes a change from state to state will be:
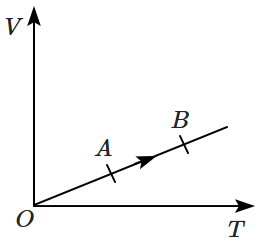
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)