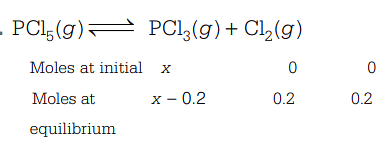KEAM SERIES Chemistry
Equilibrium Constant
6 previous year questions.
Volume: 6 Ques
Yield: Medium
High-Yield Trend
4
2026 1
2016 1
2015 Chapter Questions 6 MCQs
01
PYQ 2015
medium
chemistry ID: keam-201
The equilibrium constant for the equilibrium at a particular temperature is . The number of moles of that must be taken in a one litre flask at the same temperature to obtain a concentration of mole of chlorine at equilibrium is
1
2.2
2
2
3
1.8
4
0.2
02
PYQ 2016
hard
chemistry ID: keam-201
The equilibrium constant for the following reactions and are K1, K2 and K3 respectively. The equilibrium constant (K) for the reaction
1
2
3
4
03
PYQ 2026
medium
chemistry ID: keam-202
At equilibrium, , and at 800 K in a closed vessel. The for the equilibrium at 800 K is}
1
1.0
2
0.3
3
2.0
4
4.0
5
3.0
04
PYQ 2026
medium
chemistry ID: keam-202
Concenttration of and are and respectively. Equilibrium concentration of XO (Kc = 0.5)
05
PYQ 2026
medium
chemistry ID: keam-202
is taken in a closed container at K and at a pressure of bar. When a certain amount of carbon is added to the system at equilibrium some of is converted into and the equilibrium pressure is bar. What is the value at K for the equilibrium ?
1
bar
2
bar
3
bar
4
bar
5
bar
06
PYQ 2026
medium
chemistry ID: keam-202
For the equilibrium, , the value of the equilibrium constant is atm at K. The value of for the equilibrium at the same temperature is L atm mol K
1
mol L
2
mol L
3
mol L
4
mol L
5
mol L