NEET Chemistry
Phenols Preparation Properties
Low Priority
Consistency: 35%
Weightage: 7 / 20 Yrs
High-Yield Trend
1
2022 2
2020 1
2019 1
2017 1
2015 1
2010 1
2009 Questions 8 MCQs
01
PYQ 2009
easy
chemistry ID: neet-200
Phenol in a set of reactions (as captured below) yielded a product Z.
The structure of the product Z would be:
| 1. | Toluene | 2. | Benzaldehyde |
| 3. | Benzoic acid | 4. | Benzene |
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2010
easy
chemistry ID: neet-201
The most acidic compound among the following is:
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2015
easy
chemistry ID: neet-201
The reaction of phenol with chloroform in the presence of dilute sodium hydroxide finally introduces a functional group X. The X is:
| 1. | -CH2Cl | 2. | -COOH |
| 3. | -CHCl2 | 4. | -CHO |
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2017
easy
chemistry ID: neet-201
The most acidic compound among the following is:
| 1. | 2. | |
|
| 3. | |
4. | |
1
(1)
2
(2)
3
(3)
4
(4)
05
PYQ 2019
easy
chemistry ID: neet-201
The structure of intermediate A in the following reaction is:
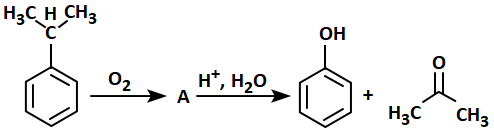
| 1. | 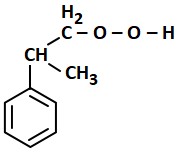 |
2. | 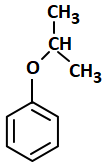 |
| 3. | 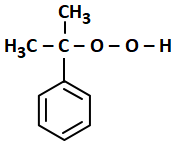 |
4. | 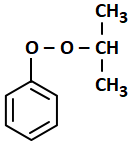 |
1
(1)
2
(2)
3
(3)
4
(4)
06
PYQ 2020
easy
chemistry ID: neet-202
The most reactive compound among the following for electrophilic aromatic substitution is:
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
07
PYQ 2020
easy
chemistry ID: neet-202
The most acidic compound among the following is:
| 1. | 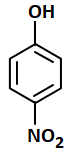 |
2. | 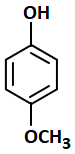 |
| 3. | 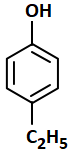 |
4. | 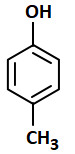 |
1
(1)
2
(2)
3
(3)
4
(4)
08
PYQ 2022
easy
chemistry ID: neet-202
Given below are two statements:
In the light of the above statements, choose the most appropriate answer from the options given below:
| I: | The acidic strength of monosubstituted nitrophenol is higher than phenol because of electron withdrawing nitro group. |
| II: | o-Nitrophenol, m-nitrophenol and p-nitrophenol will have same acidic strength as they have one nitro group attached to the phenolic ring. |
| 1. | I is incorrect but II is correct. |
| 2. | Both I and II are correct. |
| 3. | Both I and II are incorrect. |
| 4. | I is correct but II is incorrect. |
1
(1)
2
(2)
3
(3)
4
(4)