NEET Chemistry
Arrhenius Equation
Low Priority
Consistency: 35%
Weightage: 7 / 20 Yrs
High-Yield Trend
3
2024 1
2023 1
2021 1
2020 2
2013 1
2010 1
2008 Questions 10 MCQs
01
PYQ 2008
easy
chemistry ID: neet-200
The rate constants k1 and k2 for two different reactions are 1016. e-2000/T and 1015. e-1000/T, respectively. The temperature at which k1= k2 is:
1.
2.
3.
4.
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2010
easy
chemistry ID: neet-201
For an endothermic reaction, the energy of activation is Ea, and the enthalpy of reaction is ΔH (both of these in kJ/mol). The minimum value of Ea will be:
1. Less than H
2. Equal to H
3. More than H
4. Equal to zero
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2013
easy
chemistry ID: neet-201
What is the activation energy for a reaction if its rate doubles when the temperature is raised from 20oC to 35oC? (R = 8.314 J mol–1 K–1)
| 1. | 269 kJ mol–1 | 2. | 34.7 kJ mol–1 |
| 3. | 15.1 kJ mol–1 | 4. | 342 kJ mol–1 |
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2013
easy
chemistry ID: neet-201
A reaction having equal energies of activation for forward and reverse reaction has:
1. ΔG = 0
2. ΔH = 0
3. ΔH = ΔG = ΔS = 0
4. ΔS = 0
1
(1)
2
(2)
3
(3)
4
(4)
05
PYQ 2020
easy
chemistry ID: neet-202
What does ZAB represent in the collision theory of chemical reactions?
| 1. | The fraction of molecules with energies greater than Ea |
| 2. | The collision frequency of reactants, A and B |
| 3. | Steric factor |
| 4. | The fraction of molecules with energies equal to Ea |
1
(1)
2
(2)
3
(3)
4
(4)
06
PYQ 2021
easy
chemistry ID: neet-202
The slope of Arrhenius Plot (ln k v/s ) of the first-order reaction is . The value of Ea of the reaction is:
[Given R = 8.314 JK–1 mol–1]
| 1. | 166 kJ mol–1 | 2. | –83 kJ mol–1 |
| 3. | 41.5 kJ mol–1 | 4. | 83.0 kJ mol–1 |
1
(1)
2
(2)
3
(3)
4
(4)
07
PYQ 2023
hard
chemistry ID: neet-202
Given below are two statements:
| Assertion (A): | A reaction can have zero activation energy. |
| Reason (R): | The minimum amount of energy required by reactant molecules so that their energy becomes equal to threshold value, is called activation energy. |
| 1. | (A) is False but (R) is True. |
| 2. | Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 3. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 4. | (A) is True but (R) is False. |
1
(1)
2
(2)
3
(3)
4
(4)
08
PYQ 2024
easy
chemistry ID: neet-202
Activation energy of any chemical reaction can be calculated if one knows the value of:
| 1. | Probability of collision. |
| 2. | Orientation of reactant molecules during collision. |
| 3. | Rate constant at two different temperatures. |
| 4. | Rate constant at standard temperature. |
1
(1)
2
(2)
3
(3)
4
(4)
09
PYQ 2024
easy
chemistry ID: neet-202
Which plot of In k vs is consistent with Arrhenius equation?
| 1. | 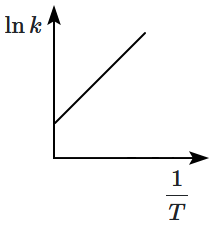 |
2. | 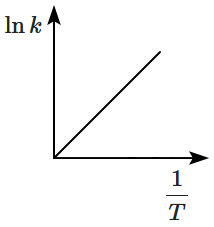 |
| 3. | 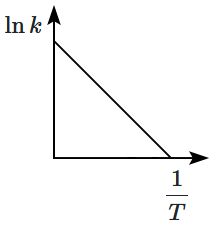 |
4. | 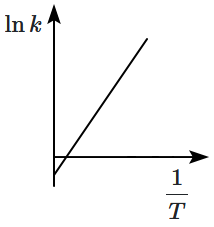 |
1
(1)
2
(2)
3
(3)
4
(4)
10
PYQ 2024
medium
chemistry ID: neet-202
The rate of a reaction quadruples when temperature changes from 27°C to 57°C. Calculate the energy of activation.
Given R = 8.314 J K–1 mol–1, log 4 = 0.6021
Given R = 8.314 J K–1 mol–1, log 4 = 0.6021
| 1. | 380.4 kJ/mol | 2. | 3.80 kJ/mol |
| 3. | 3804 kJ/mol | 4. | 38.04 kJ/mol |
1
(1)
2
(2)
3
(3)
4
(4)