NEET Chemistry
Hybridisation Structure Of Carbon Compounds
Low Priority
Consistency: 35%
Weightage: 7 / 20 Yrs
High-Yield Trend
1
2024 1
2023 1
2019 1
2013 1
2011 1
2008 1
2006 Questions 7 MCQs
01
PYQ 2006
easy
chemistry ID: neet-200
The correct order regarding the electronegativity of hybrid orbitals of carbon is:
1.
2.
3.
4.
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2008
easy
chemistry ID: neet-200
The following compounds are given for reference
(1) CH3-CH2–
(2) H2C=CH–
(3) HC≡C–
The increasing order of basic strength is:
1. (2)>(1)>(3)
2. (3) >(2) >(1)
3. (1) >(3) >(2)
4. (1) >(2)>(3)
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2011
easy
chemistry ID: neet-201
The correct order of increasing bond length of C-H, C-O, C-C and C=C is:
1.
2.
3.
4.
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2013
medium
chemistry ID: neet-201
The radical, 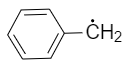 is aromatic because it has:
is aromatic because it has:
1. 7 p-orbitals and 6 electrons
2. 7 p-orbitals and 7 electrons
3. 6 p-orbitals and 7 electrons
4. 6 p-orbitals and 6 electrons
1. 7 p-orbitals and 6 electrons
2. 7 p-orbitals and 7 electrons
3. 6 p-orbitals and 7 electrons
4. 6 p-orbitals and 6 electrons
1
(1)
2
(2)
3
(3)
4
(4)
05
PYQ 2019
easy
chemistry ID: neet-201
The number of sigma () and pi () bonds in pent-3-en-1-yne are:
1. 13 bonds and no bond
2. 10 bonds and 3 bonds
3. 8 bonds and 5 bonds
4. 11 bonds and 2 bonds
1
(1)
2
(2)
3
(3)
4
(4)
06
PYQ 2023
easy
chemistry ID: neet-202
The number of bonds, bonds and lone pair of electrons in pyridine, respectively, are:
1. 12, 2, 1
2. 11, 2, 0
3. 12, 3, 0
4. 11, 3, 1
1. 12, 2, 1
2. 11, 2, 0
3. 12, 3, 0
4. 11, 3, 1
1
(1)
2
(2)
3
(3)
4
(4)
07
PYQ 2024
easy
chemistry ID: neet-202
Match List-I with List-II.
Choose the correct answer from the options given below:
| List-I (Molecule) | List-II (Number and types of bond/s between two carbon atoms) | ||
| A. | Ethane | I. | |
| B. | Ethene | II. | |
| C. | Carbon molecule, | III. | |
| D. | Ethyne | IV. |
| 1. | A-IV, B-III, C-II, D-I | 2. | A-III, B-IV, C-II, D-I |
| 3. | A-III, B-IV, C-I, D-II | 4. | A-I, B-IV, C-II, D-III |
1
(1)
2
(2)
3
(3)
4
(4)