Chemical Properties
High-Yield Trend
Questions 15 MCQs
The correct order of the mobility of the alkali metal ions in aqueous solution is:
1.
2.
3.
4.
The correct order of increasing thermal stability of K2CO3, MgCO3, CaCO3, and BeCO3 is:
1.
2.
3.
4.
In which of the following the hydration energy is higher than the lattice energy?
1.
2.
3.
4.
1. CsH > RbH > KH > NaH > LiH
2. KH > NaH > LiH > CsH > RbH
3. NaH > LiH > KH > RbH > CsH
4. LiH > NaH > KH > RbH > CsH
In the case of alkali metals, the covalent character decreases in the order:
1. MCl>Ml>MBr>MF
2. MF>MCl>MBr>Ml
3. MF>MCl>Ml>MBr
4. Ml>MBr>MCl>MF
A compound that has higher hydration enthalpy than the lattice enthalpy is:
1.
2.
3.
4.
Among the given options, what is the property of the alkaline earth metals that increases with their atomic number?
1. Solubility of their hydroxides in water
2. Solubility of their sulphates in water
3. Ionization energy
4. Electro-negativity
Which one of the following compounds is a peroxide?
1.
2.
3.
4.
Which one of the following alkali metals forms only the normal oxide M2O on heating in air?
1. Rb
2. K
3. Li
4. Na
A compound that releases CO2 most easily upon heating is?
1.
2.
3.
4.
Compound among the following that can be used as the halide component in the Friedel-Crafts reaction :
1. Chlorobenzene
2. Bromobenzene
3. Chloroethene
4. Isopropyl chloride
A compound that does not undergo SN1 reaction with OH- is:
| 1. | 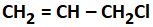 |
2. | 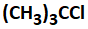 |
| 3. | 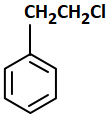 |
4. | 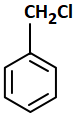 |
| 1. | A racemic mixture shows zero optical rotation |
| 2. | SN1 reaction yields a 1:1 mixture of both enantiomers |
| 3. | The product obtained by SN2 reaction of haloalkane having chirality at the reactive site shows the inversion of configuration |
| 4. | Enantiomers are superimposable mirror images on each other |
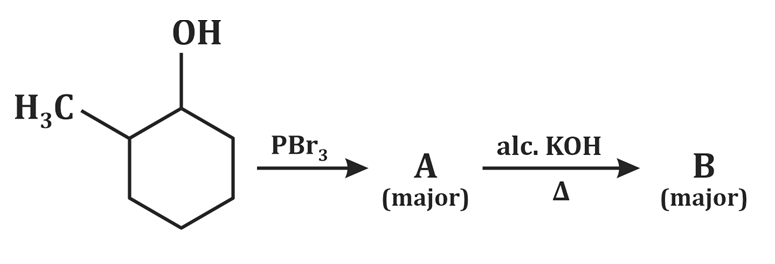
| 1. | 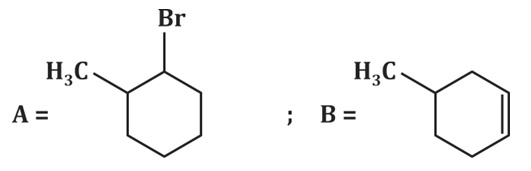 |
| 2. | 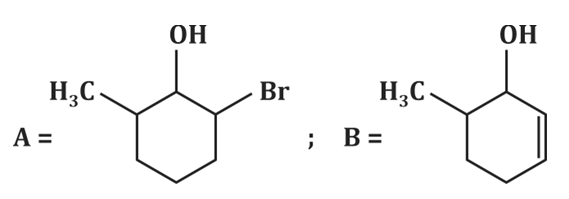 |
| 3. | 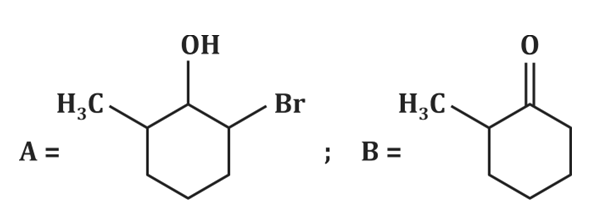 |
| 4. | 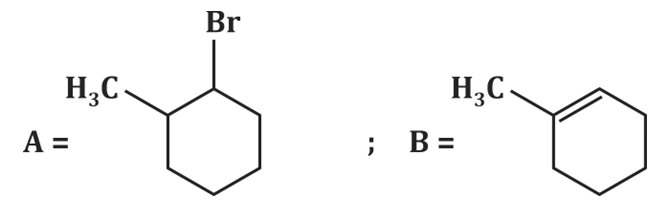 |
1.
2.
3.
4.