Amines Preparation Properties
High-Yield Trend
Questions 17 MCQs
Propanoic acid gives a series of reactions as given below.
The structure of D would be:
1.
2.
3.
4.
Which of the following compounds has a higher basic character than aniline?
1. Diphenylamine
2. Triphenylamine
3. p-nitroaniline
4. Benzylamine
What is the product formed in the below reaction?
| 1. | 2. | ||
| 3. | 4. |
The reagent that is used to convert acetamide into methyl amine is:
1. NaOH-Br2
2. Soda lime
3. Hot conc. H2SO4
4. PCl5
The incorrect statement among the following regarding the primary amine is:
| 1. | Alkyl amines are stronger bases than aryl amines |
| 2. | Alkyl amines react with nitrous acid to produce alcohols |
| 3. | Aryl amines react with nitrous acid to produce phenols |
| 4. | Alkyl amines are stronger bases than ammonia |
The number of structural isomers possible from the molecular formula C3H9N are:
| 1. | Four (4) | 2. | Five (5) |
| 3. | Two (2) | 4. | Three (3) |
Mark the reaction that does not yield aniline from the given options:
| 1. | Hydrolysis of phenyl isocyanide with an acidic solution |
| 2. | Degradation of benzamide with bromine in alkaline solution |
| 3. | Reduction of nitrobenzene with H2/Pd in ethanol |
| 4. | Treatment of potassium salt of phthalimide with chlorobenzene followed by hydrolysis in an aqueous NaOH solution |
What is the name of the following reaction?
1. Friedel-Crafts reaction
2. Perkins reaction
3. Acetylation reaction
4. Schotten-Baumann reaction
The correct increasing order of basic strength of the given molecules is:
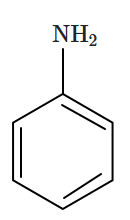 |
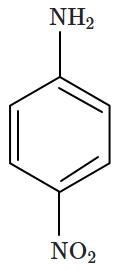 |
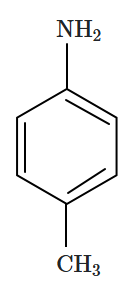 |
| (I) | (II) | (III) |
1. III < I < II
2. III < II < I
3. II < I < III
4. II < III < I
Mark the correct reaction among the following to convert acetamide to methenamine:
1. Hoffmann bromamide reaction
2. Stephens reaction
3. Gabriels phthalimide synthesis
4. Carbylamine reaction
The nitration of aniline in a strong acidic medium results in the formation of m-nitroaniline because:
| 1. | In spite of substituents, the nitro group always goes to only the m-position. |
| 2. | In electrophilic substitution reactions, the amino group is meta-directive. |
| 3. | In the absence of substituents, the nitro group always goes to only m-position. |
| 4. | In an acidic (strong) medium, aniline is present as an anilinium ion. |
1. CH3NH2 > (CH3)2NH > (CH3)3N
2. (CH3)2NH > CH3NH2 > (CH3)3N
3. (CH3)3N > CH3NH2 > (CH3)2NH
4. (CH3)2NH > (CH3)3N > CH3NH2
| 1. | Ethylamine | 2. | Methylamine |
| 3. | Propylamine | 4. | Aniline |
| 1. | 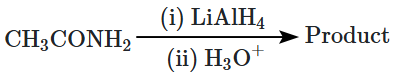 |
| 2. | 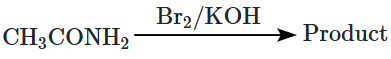 |
| 3. | 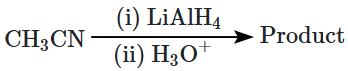 |
| 4. | 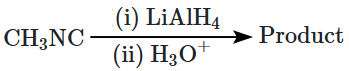 |
| Statement I: | Aniline does not undergo Friedel-Crafts alkylation reaction. |
| Statement II: | Aniline cannot be prepared through Gabriel synthesis. |
| 1. | Both Statement I and Statement II are incorrect. |
| 2. | Statement I is correct and Statement II is incorrect. |
| 3. | Statement I is incorrect and Statement II is correct. |
| 4. | Both Statement I and Statement II are correct. |
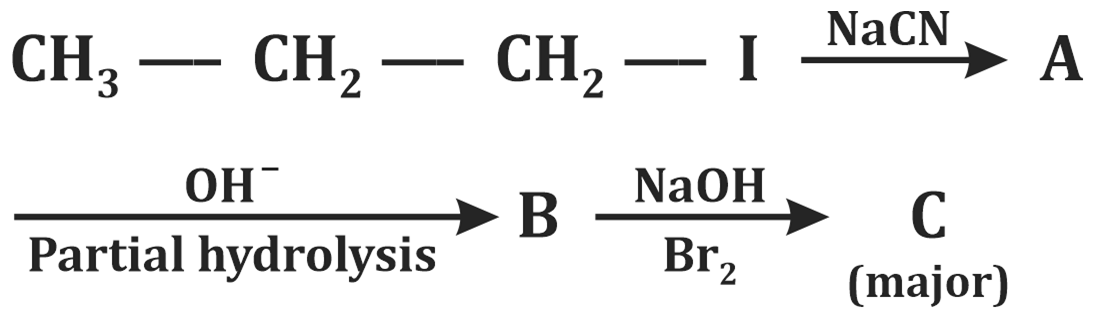
| 1. | Butylamine | 2. | Butanamide |
| 3. | Bromobutanoic acid | 4. | Propylamine |
| 1. | N-ethylethanamine > ethanamine > N -methylaniline > benzenamine |
| 2. | benzenamine > ethanamine > N -methylaniline >N-ethylethanamine |
| 3. | N -methylaniline > benzenamine > ethanamine >N-ethylethanamine |
| 4. | N-ethylethanamine > ethanamine > benzenamine >N-methylaniline |