Aldehydes Ketones Preparation Properties
High-Yield Trend
Questions 21 MCQs
A carbonyl compound reacts with hydrogen cyanide to form cyanohydrins, which form a racemic mixture of \alpha -hydroxy acid on hydrolysis. The carbonyl compound is:
1. Acetaldehyde
2. Acetone
3. Diethyl ketone
4. Formaldehyde
The self-condensation product of two moles of ethyl acetate in the presence of sodium ethoxide yield is:
1. Ethyl butyrate
2. Acetoacetic ester
3. Methyl acetoacetate
4. Ethyl propionate
Reduction of aldehydes and ketones into hydrocarbons using amalgam and conc. HCl is called:
1. Clemmensen reduction
2. Cope reduction
3. Dow reduction
4. Wolff-Kishner reduction
A compound with the molecular formula C5H10 that yields acetone on ozonolysis is:
1. 2-Methyl-2-butene
2. 2-Methyl-1-butene
3. Cyclopentane
4. 3-Methyl-1-butene
The product formed in aldol condensation is:
| 1. | A -hydroxy acid |
| 2. | A -hydroxy aldehyde or a -hydroxy ketone |
| 3. | An -hydroxy aldehyde or ketone |
| 4. | An - unsaturated ester |
1. Alkene
2. Amine
3. Ketone
4. Alkane
The correct order of decreasing acid strength of
trichloroacetic acid (A), trifluoroacetic acid (B), acetic acid (C), and formic acid (D) is:
1. B > A > D > C
2. B > D > C > A
3. A > B > C > D
4. A > C > B >D
The esters that get hydrolyzed most easily under alkaline conditions is?
| 1. | |
| 2. | |
| 3. | |
| 4. |
In the below reaction, the structure of "A" is:
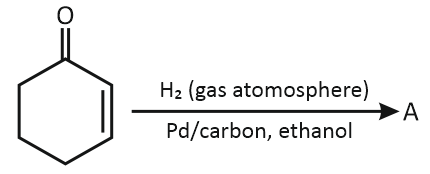
| 1. | 2. | 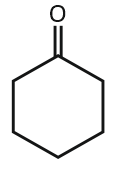 |
|
| 3. | 4. |
What is the correct order of the carboxylic acids' strength?
| I. | II. | III. |
| 1. | I > II > III | 2. | II > III > I |
| 3. | III > II > I | 4. | II > I > III |
A, X, Y, and Z in the above-mentioned reaction are:
| A | X | Y | Z | |
| 1. | Methoxy- methane |
Ethanol | Ethanoic acid | Semicarbazone |
| 2. | Ethanal | Ethanol | But - 2 - enal | semicarbazone |
| 3. | Ethanol | Acetaldehyde | Butanone | Hydrazone |
| 4. | Methoxy- methane |
Ethanoic acid | Acetate | Hydrazine |
1. Dichloromethyl cation
2. Formyl cation
3. Dichloromethyl anion
4. Dichlorocarbene (:CCl2)
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to:
| 1. | Formation of intramolecular H-bonding |
| 2. | Formation of carboxylate ion |
| 3. | More extensive association of carboxylic acid via van der waals force of attraction |
| 4. | Formation of intermolecular H-bonding |
The intermediate compound 'X' in the following chemical reaction is :
| 1. | 2. | ||
| 3. | 4. |
| Statement I | The boiling points of aldehydes and ketones are higher than hydrocarbons of comparable molecular masses because of weak molecular association in aldehydes and ketones due to dipole-dipole interactions. |
| Statement II | The boiling points of aldehydes and ketones are lower than the alcohols of similar molecular masses due to the absence of H-bonding. |
| 1. | Statement I is incorrect but Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct but Statement II is incorrect. |
The final product [C] is:
| 1. | 2. | ||
| 3. | 4. |
| 1. | 2. | ||
| 3. | 4. |
| List-I (Reaction) |
List-II (Reagents/ Condition) |
||
| A. | 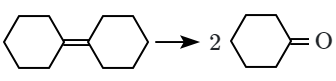 |
I. | 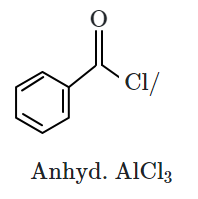 |
| B. | 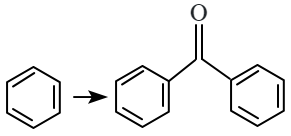 |
II. | |
| C. | 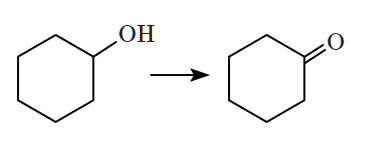 |
III. | |
| D. | 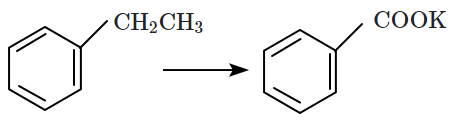 |
IV. |
| 1. | |
| 2. | |
| 3. | |
| 4. |
| 1. | Alkaline copper sulphate |
| 2. | Alkaline solution of sodium potassium tartrate (Rochelle's salt) |
| 3. | Aqueous sodium citrate |
| 4. | Aqueous copper sulphate |
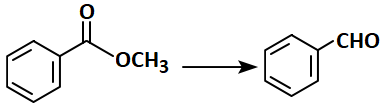
1. (i) , (ii)
2.
3. (i) , (ii)
4. (i) (ii)
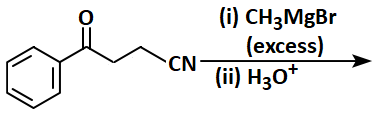
| 1. | 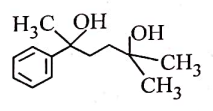 |
2. | 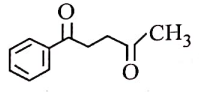 |
| 3. | 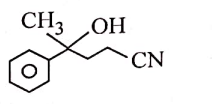 |
4. | 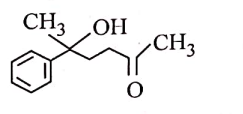 |