NEET Chemistry
Enthalpy Internal Energy
Low Priority
Consistency: 25%
Weightage: 5 / 20 Yrs
High-Yield Trend
1
2025 1
2023 1
2021 1
2012 1
2011 Questions 5 MCQs
01
PYQ 2011
easy
chemistry ID: neet-201
Given the following reaction:
→
The enthalpy change for the reaction is -869.6 kJ. The dissociation energy of the H-H bond is:
1. -869.6 kJ
2. +434.8kJ
3. +217.4kJ
4. -434.8 kJ
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2012
easy
chemistry ID: neet-201
The standard enthalpy of vaporization for water at 100 oC is 40.66 kJ mol–1. The internal energy of vaporization of water at 100 oC (in kJ mol–1) is:
(Assume water vapour behaves like an ideal gas.)
| 1. | +37.56 | 2. | –43.76 |
| 3. | +43.76 | 4. | +40.66 |
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2021
easy
chemistry ID: neet-202
For a reaction AB, enthalpy of reaction is and enthalpy of activation is . The correct potential energy profile for the reaction is:
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2023
easy
chemistry ID: neet-202
What is the correct relationship between changes in enthalpy and internal energy within the following options?
1.
2.
3.
4.
1.
2.
3.
4.
1
(1)
2
(2)
3
(3)
4
(4)
05
PYQ 2025
medium
chemistry ID: neet-202
Which of the following diagrams gives an accurate representation of the above reaction?
[R→reactants; P→products]
| 1. | 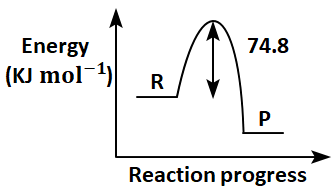 |
2. | 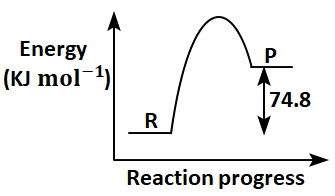 |
| 3. | 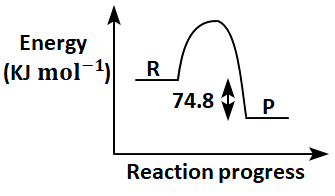 |
4. | 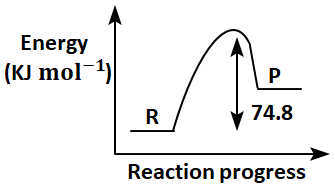 |
1
(1)
2
(2)
3
(3)
4
(4)