NEET Chemistry
Diazonium Salts Preparation Properties Uses
Low Priority
Consistency: 35%
Weightage: 7 / 20 Yrs
High-Yield Trend
1
2025 1
2023 2
2022 1
2021 1
2013 1
2010 1
2008 Questions 8 MCQs
01
PYQ 2008
easy
chemistry ID: neet-200
The structure of C in the below-mentioned reaction is:
| 1. | |
| 2. | |
| 3. | |
| 4. |
1
(1)
2
(2)
3
(3)
4
(4)
02
PYQ 2010
easy
chemistry ID: neet-201
Aniline gives a set of the following reactions that yielded a coloured product 'Y':
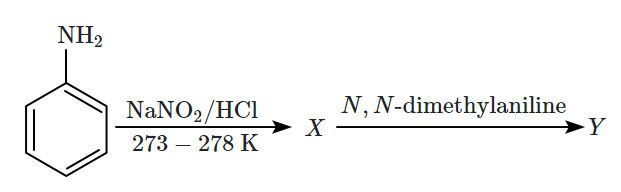
The structure of 'Y' is :
| 1. | 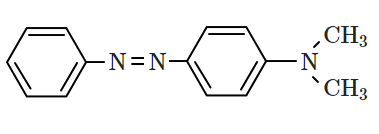 |
| 2. | 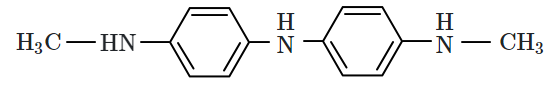 |
| 3. | 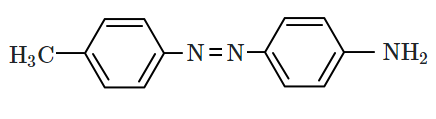 |
| 4. | 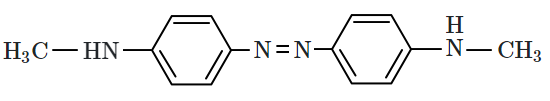 |
1
(1)
2
(2)
3
(3)
4
(4)
03
PYQ 2013
easy
chemistry ID: neet-201
"A" in the below reaction is:
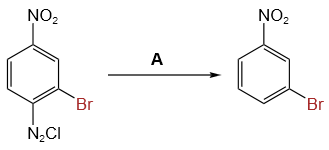
1. Cu2Cl2
1. Cu2Cl2
2. H3PO2 and H2O
3. H+ / H2O
4. HgSO4 / H2SO4
1
(1)
2
(2)
3
(3)
4
(4)
04
PYQ 2021
medium
chemistry ID: neet-202
The reagent 'R' in the given sequence of a chemical reaction is :
| 1. | HI | 2. | CuCN/KCN |
| 3. | H2O | 4. | CH3CH2OH |
1
(1)
2
(2)
3
(3)
4
(4)
05
PYQ 2022
medium
chemistry ID: neet-202
Given below are two statements:
In the light of the above statements, choose the most appropriate answer from the options given below:
| Statement I: | Primary aliphatic amines react with HNO2 to give unstable diazonium salts. |
| Statement II: | Primary aromatic amines react with HNO2 to form diazonium salts which are stable even above 300 K. |
| 1. | Statement I is incorrect but Statement II is correct |
| 2. | Both Statement I and II are correct |
| 3. | Both Statement I and II are incorrect |
| 4. | Statement I is correct but Statement II is incorrect |
1
(1)
2
(2)
3
(3)
4
(4)
06
PYQ 2022
easy
chemistry ID: neet-202
The product formed from the following reaction sequence is
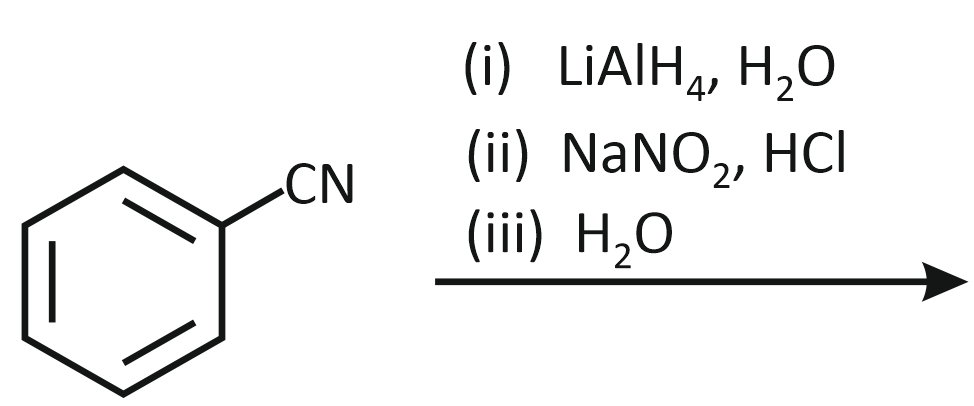
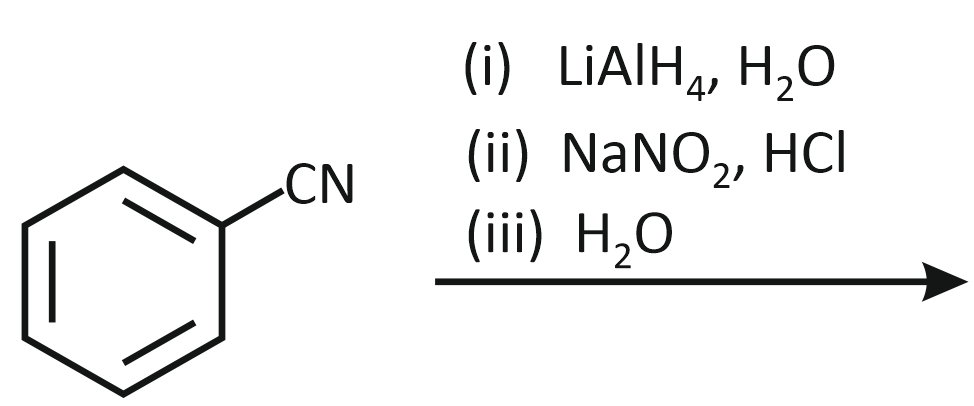
| 1. | 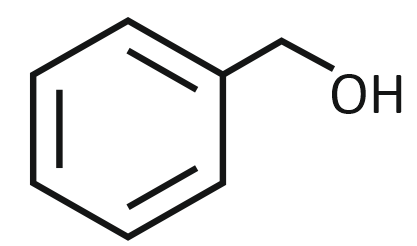 |
2. | 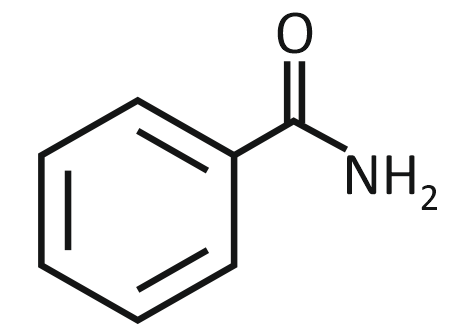 |
| 3. | 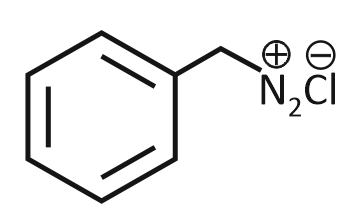 |
4. | 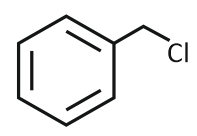 |
1
(1)
2
(2)
3
(3)
4
(4)
07
PYQ 2023
medium
chemistry ID: neet-202
Identify the product in the following reaction:
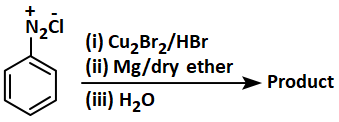
| 1. | 2. | ||
| 3. | 4. |
1
(1)
2
(2)
3
(3)
4
(4)
08
PYQ 2025
easy
chemistry ID: neet-202
Given below are two statements:
| Statement I: | Benzenediazonium salt is prepared by the reaction of aniline with nitrous acid at It decomposes easily in the dry state. |
| Statement II: | Insertion of iodine into the benzene ring is difficult, and hence iodobenzene is prepared through the reaction of benzenediazonium salt with |
| 1. | Statement I is correct; Statement II is incorrect |
| 2. | Statement I is incorrect; Statement II is correct |
| 3. | Both Statement I and Statement II are correct |
| 4. | Both Statement I and Statement II are incorrect |
1
(1)
2
(2)
3
(3)
4
(4)