JEE-ADVANCED SERIES Chemistry
Biomolecules
6 previous year questions.
Volume: 6 Ques
Yield: Medium
High-Yield Trend
2
2025 1
2024 1
2020 1
2019 1
2011 Chapter Questions 6 MCQs
01
PYQ 2011
medium
chemistry ID: jee-adva
The following carbohydrate is

1
a ketohexose
2
an aldohexose
3
an α-furanose
4
an α-pyranose
02
PYQ 2019
medium
chemistry ID: jee-adva
Which of the following statements) is (are) true?
1
Monosaccharides cannot be hydrolysed to give polyhydroxy aldehydes and ketones
2
Oxidation of glucose with bromine water gives glutamic acid
3
Hydrolysis of sucrose gives dextrorotatory glucose and laevorotatory fructose
4
The two six-membered cyclic hemiacetal forms of D-(+)-glucose are called anomers
03
PYQ 2020
medium
chemistry ID: jee-adva
The structure of a peptide is given below:

If the absolute values of the net charge of the peptide at , and are , and , respectively, then what is ?

If the absolute values of the net charge of the peptide at , and are , and , respectively, then what is ?
04
PYQ 2024
medium
chemistry ID: jee-adva
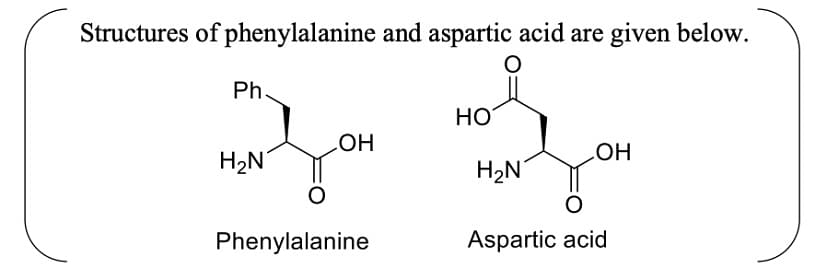
Aspartame, an artificial sweetener, is a dipeptide aspartyl phenylalanine methyl ester. The structure of aspartame is

1

2

3

4

05
PYQ 2025
medium
chemistry ID: jee-adva
A linear octasaccharide (molar mass = 1024 g mol ) on complete hydrolysis produces three monosaccharides: ribose, 2-deoxyribose and glucose. The amount of 2-deoxyribose formed is 58.26 % (w/w) of the total amount of the monosaccharides produced in the hydrolyzed products. The number of ribose unit(s) present in one molecule of octasaccharide is _____.
Use: Molar mass (in g mol ): ribose = 150, 2-deoxyribose = 134, glucose = 180; Atomic mass (in amu): H = 1, O = 16
Use: Molar mass (in g mol ): ribose = 150, 2-deoxyribose = 134, glucose = 180; Atomic mass (in amu): H = 1, O = 16
06
PYQ 2025
easy
chemistry ID: jee-adva
A linear octasaccharide (molar mass = 1024 g mol ) on complete hydrolysis produces three monosaccharides: ribose, 2-deoxyribose and glucose. The amount of 2-deoxyribose formed is 58.26 % (w/w) of the total amount of the monosaccharides produced in the hydrolyzed products. The number of ribose unit(s) present in one molecule of octasaccharide is _____.
Use: Molar mass (in g mol ): ribose = 150, 2-deoxyribose = 134, glucose = 180; Atomic mass (in amu): H = 1, O = 16
Use: Molar mass (in g mol ): ribose = 150, 2-deoxyribose = 134, glucose = 180; Atomic mass (in amu): H = 1, O = 16



