JEE-ADVANCED SERIES Chemistry
Laws Of Thermodynamics
1 previous year questions.
Volume: 1 Ques
Yield: Medium
High-Yield Trend
1
2024 Chapter Questions 1 MCQs
01
PYQ 2024
easy
chemistry ID: jee-adva
Consider the following volume−temperature (V−T) diagram for the expansion of 5 moles of an ideal monoatomic gas.
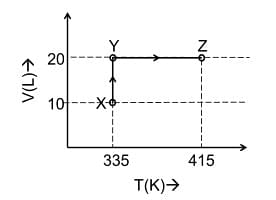
Considering only P-V work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence X→Y→Z is ______.
[Use the given data: Molar heat capacity of the gas for the given temperature range, CV,m = 12 J K−1mol−1 and gas constant, R=8.3 JK−1 mol−1]

Considering only P-V work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence X→Y→Z is ______.
[Use the given data: Molar heat capacity of the gas for the given temperature range, CV,m = 12 J K−1mol−1 and gas constant, R=8.3 JK−1 mol−1]