JEE-ADVANCED SERIES Chemistry
Kinetics Equations
2 previous year questions.
Volume: 2 Ques
Yield: Medium
High-Yield Trend
1
2020 1
2017 Chapter Questions 2 MCQs
01
PYQ 2017
medium
chemistry ID: jee-adva
In a bimolecular reaction, the steric factor was experimentally determined to be The correct option(s) among the following is(are)
1
The activation energy of the reaction is unaffected by the value of the steric factor
2
Experimentally determined value of frequency factor is higher than that predicted by Arrhenius equation
3
Since P = 4.5, the reaction will not proceed unless an effective catalyst is used
4
The value of frequency factor predicted by Arrhenius equation is higher than that determined experimentally
02
PYQ 2020
medium
chemistry ID: jee-adva
If the distribution of molecular speeds of a gas is as per the figure shown below, then the ratio of the most probable, the average, and the root mean square speeds, respectively, is
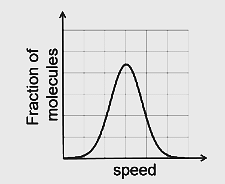

1
2
3
4