JEE-ADVANCED SERIES Chemistry
Structure Of Atom
23 previous year questions.
Volume: 23 Ques
Yield: High
High-Yield Trend
2
2024 2
2023 1
2002 3
2001 1
1998 1
1996 1
1992 1
1989 4
1988 3
1985 3
1984 1
1983 Chapter Questions 23 MCQs
01
PYQ 1983
medium
chemistry ID: jee-adva
Any p-orbital can accommodate upto
1
four electrons
2
six electrons
3
two electrons with parallel spins
4
two electrons with opposite spins
02
PYQ 1984
medium
chemistry ID: jee-adva
An isotone of is
1
2
3
4
03
PYQ 1984
medium
chemistry ID: jee-adva
Which electronic level would allow the hydrogen atom to absorb a photon but not to emit a photon?
1
3s
2
2p
3
2s
4
1s
04
PYQ 1984
medium
chemistry ID: jee-adva
An isotope of is
1
2
3
4
05
PYQ 1985
easy
chemistry ID: jee-adva
The increasing order (lowest first) for the values of e/m (charge/mass) for electron (e), proton (p), neutron (n) and alpha particle is
1
e, p, n,
2
n, p, e
3
n, p, , e
4
n, p, e
06
PYQ 1985
easy
chemistry ID: jee-adva
Bohr's model can explain
1
the spectrum of hydrogen atom only
2
spectrum of an atom or ion containing one electron only
3
the spectrum of hydrogen molecule
4
the solar spectrum
07
PYQ 1985
medium
chemistry ID: jee-adva
Electromagnetic radiation with maximum wavelength is
1
ultraviolet
2
radio wave
3
X-ray
4
infrared
08
PYQ 1988
easy
chemistry ID: jee-adva
The outermost electronic configuration of the most electronegative element is
1
2
3
4
09
PYQ 1988
easy
chemistry ID: jee-adva
The atomic nucleus contains
1
protons
2
neutrons
3
electrons
4
photons
10
PYQ 1988
medium
chemistry ID: jee-adva
The orbital diagram in which Aufbau principle is violated is
1

2

3

4

11
PYQ 1988
medium
chemistry ID: jee-adva
The wavelength of a spectral line for an electronic transition is inversely related to
1
the number of electrons undergoing the transition
2
the nuclear charge of the atom
3
the difference in the energy of the energy levels involved in the transition
4
the velocity of the electron undergoing the transition
12
PYQ 1989
medium
chemistry ID: jee-adva
The ground state electronic configuration of chromium is
1
2
3
4
13
PYQ 1992
medium
chemistry ID: jee-adva
Which of the following does not characterise X-rays ?
1
The radiation can ionise gases
2
It causes ZnS to fluoresce
3
Deflected by electric and magnetic fields
4
Have wavelengths shorter than ultraviolet rays
14
PYQ 1996
easy
chemistry ID: jee-adva
Which of the following has the maximum number of unpaired electrons ?
1
2
3
4
15
PYQ 1998
medium
chemistry ID: jee-adva
Which of the following statement (s) is (are) correct ?
1
The electronic configuration of Cr is [Ar] (atomic number of Cr = 24)
2
The magnetic quantum number may have a negative value
3
In silver atom, 23 electrons have a spin of one type and 24 of the opposite type, (atomic number of Ag = 47)
4
The oxidation state of nitrogen in is -3
16
PYQ 2001
medium
chemistry ID: jee-adva
The number of nodal planes in a orbital is
1
one
2
two
3
three
4
zero
17
PYQ 2001
medium
chemistry ID: jee-adva
The wavelength associated with a golf ball weighing 200 g and moving at a speed of 5 m/h is of the order
1
2
3
4
18
PYQ 2001
medium
chemistry ID: jee-adva
The quantum numbers and for the electron spin represent
1
rotation of the electron in clockwise and anti-clockwise direction respectively
2
rotation of the electron in anti-clockwise and clockwise direction respectively
3
magnetic moment of the electron pointing up and down respectively
4
two quantum mechanical spin states which have no classical analogue
19
PYQ 2002
medium
chemistry ID: jee-adva
If the nitrogen atom had electronic configuration , it would have energy lower than that of the normal ground state configuration , because the electrons would be closer to the nucleus, yet is not observed, because it violates
1
Heisenberg uncertainty principle
2
Hund's rule
3
Pauli exclusion principle
4
Bohr postulate of stationary orbits
20
PYQ 2023
medium
chemistry ID: jee-adva
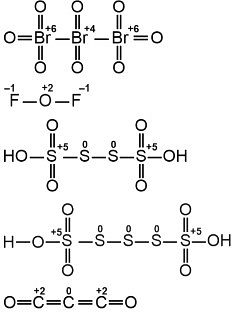
Consider the following molecules: Br3O8, F2O, H2S4O6, H2S5O6, and C3O2. Count the number of atoms existing in their zero oxidation state in each molecule. Their sum is____.
21
PYQ 2023
medium
chemistry ID: jee-adva
Consider the following molecules: Br3O8, F2O, H2S4O6, H2S5O6, and C3O2. Count the number of atoms existing in their zero oxidation state in each molecule. Their sum is____.
22
PYQ 2024
medium
chemistry ID: jee-adva
According to Bohr’s model, the highest kinetic energy is associated with the electron in the
1
first orbit of H atom
2
first orbit of He+
3
second orbit of He+
4
second orbit of Li2+
23
PYQ 2024
medium
chemistry ID: jee-adva
Among the following, the correct statement(s) for electrons in an atom is(are)
1
Uncertainty principle rules out the existence of definite paths for electrons.
2
The energy of an electron in 2s orbital of an atom is lower than the energy of an electron that is infinitely far away from the nucleus.
3
According to Bohr's model, the most negative energy value for an electron is given by n = 1, which corresponds to the most stable orbit.
4
According to Bohr's model, the magnitude of velocity of electrons increases with increase in values of n.