JEE-ADVANCED SERIES Chemistry
Stoichiometry And Stoichiometric Calculations
10 previous year questions.
Volume: 10 Ques
Yield: Medium
High-Yield Trend
5
2023 3
2021 2
2020 Chapter Questions 10 MCQs
01
PYQ 2020
medium
chemistry ID: jee-adva
Aluminium reacts with sulfuric acid to form aluminium sulfate and hydrogen. What is the volume of hydrogen gas in liters produced at and atm pressure, when of aluminium and of sulfuric acid are combined for the reaction?
(Use molar mass of aluminium as
(Use molar mass of aluminium as
02
PYQ 2020
medium
chemistry ID: jee-adva
Consider the reaction sequence from to shown below. The overall yield of the major product from is . What is the amount in grams of obtained from of . (Use density of ; Molar mass of and )


03
PYQ 2021
medium
chemistry ID: jee-adva
For the following reaction scheme, percentage yields are given along the arrow:

and are mass of and , respectively (Use : Molar mass (in of and as and , respectively) The value of is ______

and are mass of and , respectively (Use : Molar mass (in of and as and , respectively) The value of is ______
04
PYQ 2021
medium
chemistry ID: jee-adva
For the following reaction scheme, percentage yields are given along the arrow:
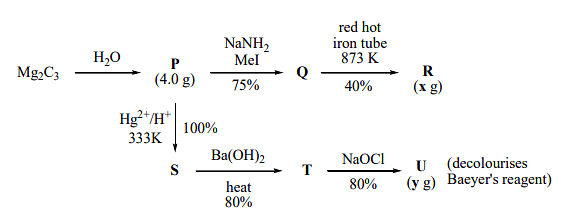
and are mass of and , respectively (Use : Molar mass (in of and as and , respectively) The value of is _______
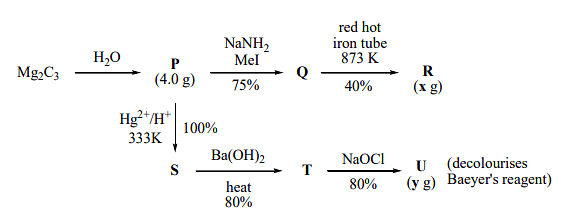
and are mass of and , respectively (Use : Molar mass (in of and as and , respectively) The value of is _______
05
PYQ 2021
easy
chemistry ID: jee-adva
For the following reaction scheme, percentage yields are given along the arrow:

and are mass of and , respectively. (Use : Molar mass (in of and as and , respectively)

and are mass of and , respectively. (Use : Molar mass (in of and as and , respectively)
06
PYQ 2023
easy
chemistry ID: jee-adva
Match the reactions (in the given stoichiometry of the reactants) in List-I with one of their products given in List-II and choose the correct option.
List-I | List-II | ||
| (P) | P2O3 + 3H2O → | (1) | P(O)(OCH3)Cl2 |
| (Q) | P4 + 3NaOH + 3H2O → | (2) | H3PO3 |
| (R) | PCl5 + CH3COOH → | (3) | PH3 |
| (S) | H3PO2 + 2H2O + 4AgNO3 → | (4) | POCl3 |
| (5) | H3PO4 |
1
P →2; Q→ 3; R →1; S →5
2
P →3; Q→ 5; R →4; S →2
3
P →5; Q→ 2; R →1; S →3
4
P →2; Q→ 3; R →4; S →5
07
PYQ 2023
easy
chemistry ID: jee-adva
In the following reactions, P, Q, R, and S are the major products.

The correct statement(s) about P, Q, R, and S is(are)

The correct statement(s) about P, Q, R, and S is(are)
1
P and Q are monomers of polymers dacron and glyptic, respectively.
2
P, Q, and R are dicarboxylic acids.
3
Compounds Q and R are the same.
4
R does not undergo aldol condensation and S does not undergo Cannizzaro reaction.
08
PYQ 2023
easy
chemistry ID: jee-adva
Match the electronic configurations in List-I with appropriate metal complex ions in List-II and choose the correct option.
[Atomic Number: Fe = 26, Mn = 25, Co = 27]
[Atomic Number: Fe = 26, Mn = 25, Co = 27]
| List-I | List-II | ||
| (P) | (1) | ||
| (Q) | (2) | ||
| (R) | (3) | ||
| (S) | (4) | ||
| (5) |
1
P →1; Q→ 4; R →2; S →3
2
P →1; Q→ 2; R →4; S →5
3
P →3; Q→ 2; R →5; S →1
4
P →3; Q→ 2; R →4; S →1
09
PYQ 2023
easy
chemistry ID: jee-adva
In the given reaction scheme, P is a phenyl alkyl ether, Q is an aromatic compound; R and S are the major products.

The correct statement about S is

The correct statement about S is
1
It primarily inhibits noradrenaline degrading enzymes
2
It inhibits the synthesis of prostaglandin.
3
It is a narcotic drug.
4
It is ortho-acetylbenzoic acid.
10
PYQ 2023
hard
chemistry ID: jee-adva
Match the reactions in List-I with the features of their products in List-II and choose the correct option
| List-I | List-II | ||
| (P) | (-)-1-Bromo-2-ethylpentane  (single enantiomer) | (1) | Inversion of configuration |
| (Q) | (-)-2-Bromopentane  (single enantiomer) | (2) | Retention of configuration |
| (R) | (-)-3- Bromo-3-methylhexane  (single enantiometer) | (3) | Mixture of enantiomers |
| (S) |   (single enantiometer) | (4) | Mixture of structural isomers |
| (5) | Mixture of diastereomers |
1
P →1; Q→2 ; R →5; S →3
2
P →2; Q→1 ; R →3; S →5
3
P →1; Q→2; R →5; S →4
4
P →2; Q→4; R →3; S →5
