Organic Chemistry
44 previous year questions.
High-Yield Trend
Chapter Questions 44 MCQs


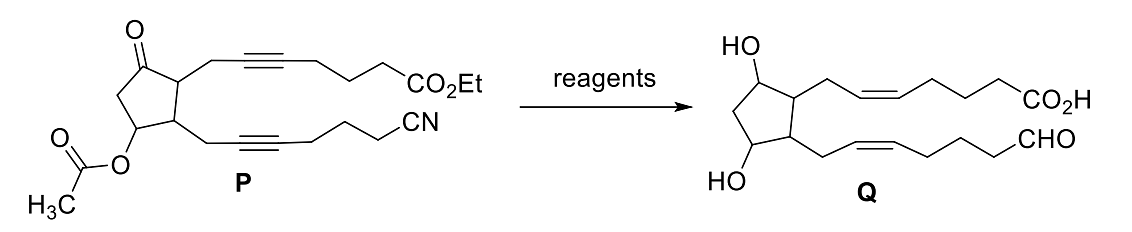
|What is the degree of unsaturation of Q?


The major products obtained from the reactions in List-II are the reactants for the named reactions mentioned in List-I. Match List-I with List-II and choose the correct option.

P →2; Q→4 ; R →1; S →3
P →1; Q→3 ; R →5; S →2
P →3; Q→2 ; R →1; S →4
P →3; Q→4 ; R →5; S →2
[ = molar conductivity,
= limiting molar conductivity,
= molar concentration,
= dissociation constant of HX]
[Use: Molar mass of urea = 60 g mol−1 ; gas constant, R = 62 L Torr K−1 mol−1; Assume, ΔmixH = 0, ΔmixV = 0]
A disaccharide X cannot be oxidised by bromine water. The acid hydrolysis of X leads to a laevorotatory solution. The disaccharide X is




The total number of sp2 hybridised carbon atoms in the major product P (a non-heterocyclic compound) of the following reaction is ___.

On decreasing the 𝑝H from 7 to 2, the solubility of a sparingly soluble salt (MX) of a weak acid (HX) increased from 10−4 mol L−1 to 10−3 mol L−1. The 𝑝Ka of HX is

In the following reactions, P, Q, R, and S are the major products.
The correct statement(s) about P, Q, R, and S is(are)
P and Q are monomers of polymers dacron and glyptal, respectively.
P, Q, and R are dicarboxylic acids.
Compounds Q and R are the same.
R does not undergo aldol condensation and S does not undergo Cannizzaro reaction.
[Use: Molar mass (in g mol–1): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5 Atoms other than C and H are considered as heteroatoms]
[Use: Molar mass (in g mol–1): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5 Atoms other than C and H are considered as heteroatoms]
[Use: Molar mass of urea = 60 g mol−1 ; gas constant, R = 62 L Torr K−1 mol−1; Assume, ΔmixH = 0, ΔmixV = 0]

[Use: Molar mass (in g mol–1): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5 Atoms other than C and H are considered as heteroatoms]
[Use: Molar mass (in g mol–1): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5 Atoms other than C and H are considered as heteroatoms]
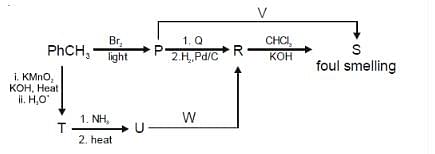
For the reaction sequence given below, the correct statement(s) is(are):
Monocyclic compounds and are the major products formed in the reaction sequences given below.
The product having the highest number of unsaturated carbon atom(s) is:




The major products obtained from the reactions in List-II are the reactants for the named reactions mentioned in List-I. Match each entry in List-I with the appropriate entry in List-II and choose the correct option. 
P → 5; Q → 4; R → 2; S → 1
Match the compounds in List-I with the appropriate observations in List-II and choose the correct option. 
P → 2; Q → 1; R → 5; S → 3
For the reaction sequence given below, the correct statement(s) is (are): 
(In the options, X is any atom other than carbon and hydrogen, and it is different in P, Q, and R.)
value of the conjugate acids of the leaving groups in and follows the order
The reaction sequence given below is carried out with 16 moles of X. The yield of the major product in each step is given below the product in parentheses. The amount (in grams) of S produced is ____. 
Use: Atomic mass (in amu): H = 1, C = 12, O = 16, Br = 80







