JEE-ADVANCED SERIES Chemistry
Redox Reactions
17 previous year questions.
Volume: 17 Ques
Yield: Medium
High-Yield Trend
2
2025 2
2023 4
2022 1
2021 2
2020 2
2014 2
2012 1
2011 1
2009 Chapter Questions 17 MCQs
01
PYQ 2009
medium
chemistry ID: jee-adva
For the reduction of ion in an aqueous solution is . Values of for some metal ions are given below The pair(s) of metals that is/are oxidised by in aqueous solution is (are)
1
and
2
and
3
and
4
and
02
PYQ 2011
medium
chemistry ID: jee-adva
Oxidation states of the metal in the minerals haematite and magnetite, respectively, are
1
II, III in haematite and III in magnetite
2
II, III in haematite and II in magnetite
3
II in haematite and II, III in magnetite
4
III in haematite and II, III in magnetite
03
PYQ 2012
medium
chemistry ID: jee-adva
Which ordering of compounds is according to the decreasing order of the oxidation state of nitrogen?
1
2
3
4
04
PYQ 2012
easy
chemistry ID: jee-adva
The reaction of white phosphorus with aqueous gives phosphine along with another phosphorus containing compound. The reaction type, the oxidation states of phosphorus in phosphine and the other product respectively are
1
redox reaction, and
2
redox reaction, and
3
disproportionation reaction, and
4
disproportionation reaction, and
05
PYQ 2014
medium
chemistry ID: jee-adva
Hydrogen peroxide in its reaction with and respectively, is acting as a
1
reducing agent, oxidising agent
2
reducing agent, reducing agent
3
oxidising agent, oxidising agent
4
oxidising agent, reducing agent
06
PYQ 2014
medium
chemistry ID: jee-adva
For the reaction: The correct statement(s) in the balanced equation is/are:
1
stoichiometric coefficient of is
2
iodide is oxidised
3
sulphur is reduced
4
is one of the products
07
PYQ 2020
medium
chemistry ID: jee-adva
In the chemical reaction between stoichiometric quantities of and in weakly basic solution, what is the number of moles of released for moles of consumed?
08
PYQ 2020
medium
chemistry ID: jee-adva
An acidified solution of potassium chromate was layered with an equal volume of amyl alcohol. When it was shaken after the addition of of , a blue alcohol layer was obtained. The blue color is due to the formation of a chromium (VI) compound ' '. What is the number of oxygen atoms bonded to chromium through only single bonds in a molecule of ?
09
PYQ 2021
easy
chemistry ID: jee-adva
A sample (5.6 g) containing iron is completely dissolved in cold dilute HCl to prepare a 250 mL of solution. Titration of 25.0 mL of this solution requires 12.5 mL of 0.03 M KMnO4 solution to reach the endpoint. Number of moles of Fe2+ present in 250 mL solution is x × 10–2 (consider complete dissolution of FeCl2). The amount of iron present in the sample is y% by weight.
(Assume: KMnO4 reacts only with Fe2+ in the solution Use: Molar mass of iron as 56 g mol–1)
10
PYQ 2022
medium
chemistry ID: jee-adva
Consider the following reaction

On estimation of bromine in of using Carius method, the amount of formed (in ) is __ [Given : Atomic mass of ]

On estimation of bromine in of using Carius method, the amount of formed (in ) is __ [Given : Atomic mass of ]
11
PYQ 2022
medium
chemistry ID: jee-adva
Consider the following reaction

On estimation of bromine in of using Carius method, the amount of formed (in ) is __ [Given : Atomic mass of ]

On estimation of bromine in of using Carius method, the amount of formed (in ) is __ [Given : Atomic mass of ]
12
PYQ 2022
medium
chemistry ID: jee-adva
The treatment of an aqueous solution of of with excess results in a brown solution along with the formation of a precipitate Passing through this brown solution gives another precipitate The amount of (in ) is ___ [Given : Atomic mass of ]
13
PYQ 2022
medium
chemistry ID: jee-adva
The treatment of an aqueous solution of of with excess results in a brown solution along with the formation of a precipitate.Passing through this brown solution gives another precipitate The amount of (in ) is ___ . [Given : Atomic mass of ]
14
PYQ 2023
easy
chemistry ID: jee-adva
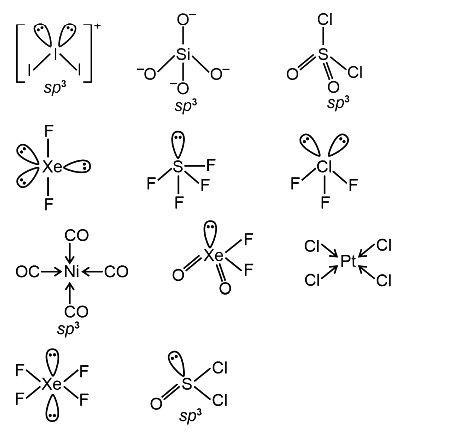
Among [I3] + , [SiO4]4-, SO2Cl2, XeF2, SF4, ClF3, Ni(CO)4, XeO2F2, [PtCl4] 2−, XeF4, and SOCl2, the total number of species having sp3 hybridised central atom is ______.
15
PYQ 2023
hard
chemistry ID: jee-adva
Among [I3] +,[SiO4]4-, SO2Cl2, XeF2, SF4, ClF3, Ni(CO)4, XeO2F2, [PtCl4] 2−, XeF4, and SOCl2, the total number of species having sp3 hybridised central atom is ______.
16
PYQ 2025
easy
chemistry ID: jee-adva
One of the products formed from the reaction of permanganate ion with iodide ion in neutral aqueous medium is:
1
2
3
4
17
PYQ 2025
medium
chemistry ID: jee-adva
In neutral medium, permanganate reacts with iodide to give:
1
,
2
,
3
,
4
,