JEE-ADVANCED SERIES Chemistry
Enthalpy Change
1 previous year questions.
Volume: 1 Ques
Yield: Medium
High-Yield Trend
1
2021 Chapter Questions 1 MCQs
01
PYQ 2021
medium
chemistry ID: jee-adva
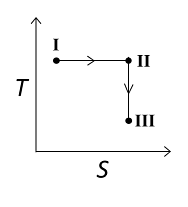
An ideal gas undergoes a reversible isothermal expansion from the state I to state II followed by a reversible adiabatic expansion from state II to state III. The correct plot(s) representing the changes from the state I to state III is(are)
(p: pressure, V: volume, T: temperature, H: enthalpy, S: entropy)
(p: pressure, V: volume, T: temperature, H: enthalpy, S: entropy)
1
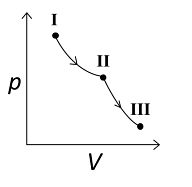
2
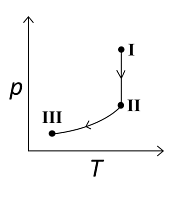
3
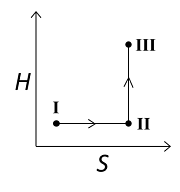
4