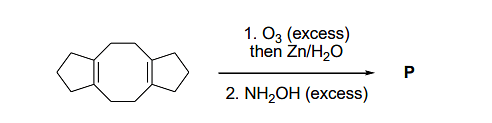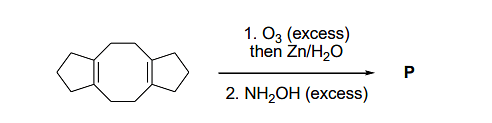JEE-ADVANCED SERIES Chemistry
Hybridisation
10 previous year questions.
Volume: 10 Ques
Yield: Medium
High-Yield Trend
1
2021 1
2010 1
1999 1
1997 1
1996 1
1992 1
1989 1
1984 1
1982 1
1981 Chapter Questions 10 MCQs
01
PYQ 1981
medium
chemistry ID: jee-adva
If a molecule , has zero dipole moment, the sigma
bonding orbitals used by M (atomic number < 21) are
1
pure p
2
sp-hybridised
3
-hybridised
4
-hybridised
02
PYQ 1982
medium
chemistry ID: jee-adva
Among the following, the linear molecule is
1
2
3
4
03
PYQ 1984
easy
chemistry ID: jee-adva
On hybridisation of one s and one p-orbital we get
1
two mutually perpendicular orbitals
2
two orbitals at
3
four orbitals directed tetrahedrally
4
three orbitals in a plane
04
PYQ 1989
medium
chemistry ID: jee-adva
The molecule which has pyramidal shape is
1
2
3
4
05
PYQ 1992
easy
chemistry ID: jee-adva
The maximum possible number of hydrogen bonds a water
molecule can form is
1
2
2
4
3
3
4
1
06
PYQ 1996
medium
chemistry ID: jee-adva
Which one of the following molecules is planar?
1
2
3
4
07
PYQ 1997
medium
chemistry ID: jee-adva
Which one of the following compounds has hybridisation?
1
2
3
4
CO
08
PYQ 1999
medium
chemistry ID: jee-adva
In the compound , the bonds is of
1
2
3
4
09
PYQ 2010
medium
chemistry ID: jee-adva
The species having pyramidal shape is
1
2
3
4
10
PYQ 2021
medium
chemistry ID: jee-adva
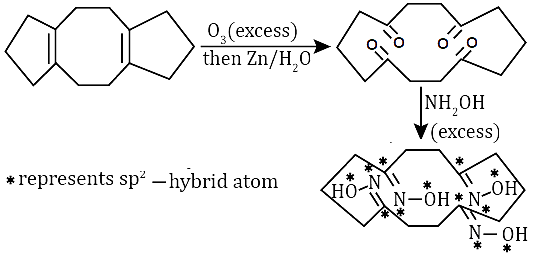
In the reaction given below, the total number of atoms having sp2 hybridization in the major product P is ___.