JEE-ADVANCED SERIES Chemistry
P Block Elements
56 previous year questions.
Volume: 56 Ques
Yield: High
High-Yield Trend
11
2022 2
2021 2
2020 1
2019 1
2017 1
2013 1
2012 1
2011 2
2009 1
2008 1
2007 4
2005 2
2004 4
2003 2
2002 Chapter Questions 56 MCQs
01
PYQ 1978
medium
chemistry ID: jee-adva
Ammonia gas can be dried by
1
cone
2
3
4
quicklime
02
PYQ 1981
medium
chemistry ID: jee-adva
HBr and HI reduce sulphuric acid, HCl can reduce and HF can reduce
1
2
3
4
None of these
03
PYQ 1983
medium
chemistry ID: jee-adva
Chlorine acts as a bleaching agent only in the presence Of
1
dry air
2
moisture
3
sunlight
4
pure oxygen
04
PYQ 1985
medium
chemistry ID: jee-adva
A gas that cannot be collected over water is
1
2
3
4
05
PYQ 1986
medium
chemistry ID: jee-adva
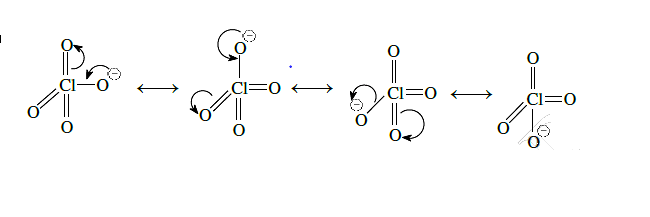
The bonds present in are
1
only ionic
2
covalent and coordinate
3
only covalent
4
covalent and inonic
06
PYQ 1987
medium
chemistry ID: jee-adva
Amongst the trihalides of nitrogen, which one is least basic?
1
2
3
4
07
PYQ 1987
medium
chemistry ID: jee-adva
Which of the following oxides of nitrogen is a coloured gas ?
1
2
NO
3
4
08
PYQ 1987
easy
chemistry ID: jee-adva
Bromine can be liberated from potassium bromide solution by the action of
1
iodine solution
2
chlorine water
3
sodium chloride
4
potassium iodide
09
PYQ 1989
medium
chemistry ID: jee-adva
Nitrogen (I) oxide is produced by
1
thermal decomposition of
2
disproportionation of
3
thermal decomposition of
4
interaction of hydroxylamine and nitrous acid
10
PYQ 1991
medium
chemistry ID: jee-adva
There is no S- S bond in
1
2
3
4
11
PYQ 1996
medium
chemistry ID: jee-adva
combines with to form The compound contains the species
1
and
2
and
3
and
4
and
12
PYQ 1996
medium
chemistry ID: jee-adva
Which of the following is the least stable and its existence is doubtful ?
1
2
3
4
13
PYQ 1996
medium
chemistry ID: jee-adva
Which one of the following oxides is neutral ?
1
CO
2
3
ZnO
4
14
PYQ 1997
medium
chemistry ID: jee-adva
Which one of the following species is not a pseudo halide?
1
2
3
4
15
PYQ 1998
medium
chemistry ID: jee-adva
White phosphorus has
1
six P-P single bonds
2
four P- P single bonds
3
four lone pairs of electrons
4
P-P-P angle of
16
PYQ 1999
medium
chemistry ID: jee-adva
One mole of calcium phosphide on reaction with excess water gives
1
one mole of phosphine
2
two moles of phosphoric acid
3
two moles of phosphine
4
one mole of phosphorus pentaoxide
17
PYQ 1999
medium
chemistry ID: jee-adva
In compounds of type , where E = B , P, As or Bi, the angles Cl-E - Cl for different E are in the order
1
B > P = As = Bi
2
B > P > As > Bi
3
B < P = As = Bi
4
B < P < As < Bi
18
PYQ 2000
medium
chemistry ID: jee-adva
Amongst and the 1one with the highest boiling point is
1
because of hydrogen bonding
2
because of higher molecular weight
3
because of hydrogen bonding
4
because of lower molecular weight
19
PYQ 2000
medium
chemistry ID: jee-adva
The number of bonds in cyclic metaphosphoric acid is
1
zero
2
two
3
three
4
four
20
PYQ 2000
medium
chemistry ID: jee-adva
Ammonia can be dried by
1
cone.
2
3
CaO
4
anhydrous
21
PYQ 2002
medium
chemistry ID: jee-adva
Polyphosphates are used as water softening agents because they
1
form soluble complexes with anionic species
2
precipitate anionic species
3
form soluble complexes with cationic species
4
precipitate cationic species
22
PYQ 2002
medium
chemistry ID: jee-adva
Identify the correct order of acidic strength of
1
2
3
4
23
PYQ 2003
medium
chemistry ID: jee-adva
For the correct choice is
1
is dibasic and reducing
2
is dibasic and non-reducing
3
is tribasic and reducing
4
is tribasic and non-reducing
24
PYQ 2003
medium
chemistry ID: jee-adva
on hydrolysis will produce
1
2
3
4
25
PYQ 2003
easy
chemistry ID: jee-adva
Graphite is a soft solid lubricant extremely difficult to melt. The reason for this anomalous behaviour is that graphite -
1
has molecules of variable molecular masses like polymers
2
has carbon atoms arranged in large plates of rings of strongly bound carbon atoms with weak interplate bonds.
3
is a non-crystalline substance.
4
is an allotropic form of carbon.
26
PYQ 2003
medium
chemistry ID: jee-adva
is
1
monobasic acid and weak Lewis acid
2
monobasic and weak Bronsted acid
3
monobasic and strong Lewis acid
4
tribasic and weak Bronsted acid
27
PYQ 2004
medium
chemistry ID: jee-adva
Which of the following has -O-O- linkage?
1
2
3
4
28
PYQ 2004
medium
chemistry ID: jee-adva
The correct statement(s) for orthoboric acid is/'are
1
It behaves as a weak acid in water due to self ionisation
2
Acidity of its aqueous solution increases upon addition of ethylene glycol
3
It has a three-dimensional structure due to hydrogen bonding
4
It is a weak electrolyte in water
29
PYQ 2005
medium
chemistry ID: jee-adva
Which of the following isomers of phosphorus is thermodynamically most stable?
1
Red
2
White
3
Black
4
Yellow
30
PYQ 2005
medium
chemistry ID: jee-adva
Which gas is evolved when is treated with concentrated ?
1
2
3
4
31
PYQ 2005
medium
chemistry ID: jee-adva
A pale blue liquid obtained by equimolar mixture of two gases at is
1
2
3
4
32
PYQ 2005
hard
chemistry ID: jee-adva
Name the structure of silicates in which three oxygen atoms of are shared is
1
pyrosilicate
2
sheet silicate
3
linear chain silicate
4
three-dimensional silicate
33
PYQ 2007
medium
chemistry ID: jee-adva
The percentage of -character in the orbitals forming bonds in is
1
25
2
33
3
50
4
75
34
PYQ 2008
medium
chemistry ID: jee-adva
Aqueous solution of on reaction with gives
1
2
3
4
35
PYQ 2009
medium
chemistry ID: jee-adva
The reaction of with leads selectively to . The , is
1
2
a mixture of
3
moist
4
in the presence of aqueous
36
PYQ 2009
medium
chemistry ID: jee-adva
In the reaction, the amine(s) X is/are
1
2
3
4
37
PYQ 2011
medium
chemistry ID: jee-adva
Extra pure can be obtained by heating
1
with
2
3
4
38
PYQ 2012
medium
chemistry ID: jee-adva
With respect to graphite and diamond, which of the statement(s) given below is/are correct?
1
Graphite is harder than diamond
2
Graphite has higher electrical conductivity than diamond.
3
Graphite has higher thermal conductivity than diamond.
4
Graphite has higher C- C bond order than diamond
39
PYQ 2013
medium
chemistry ID: jee-adva
The correct statement(s) about is/are
1
bond lengths are equal
2
thermal decomposition of is endothermic
3
is diamagnetic in nature
4
has a bent structure
40
PYQ 2017
medium
chemistry ID: jee-adva
The correct statement(s) about the oxoacids, and , is(are)
1
The conjugate base of is weaker base than
2
The central atom in both and is hybridized.
3
is more acidic than because of the resonance stabilization of its anion.
4
is formed in the reaction between and .
41
PYQ 2019
medium
chemistry ID: jee-adva
With reference to aqua regia, choose the correct option(s)
1
Aqua regia is prepared by mixing cone. HCI and cone. in 3:1 (v/v) ratio
2
Reaction of gold with aqua regia produces an anion having Au in +3 oxidation state
3
Reaction of gold with aqua regia produces in the absence of air
4
The yellow colour of aqua regia is due to the presence of NOCI and
42
PYQ 2020
medium
chemistry ID: jee-adva
Choose the correct statement(s) among the following
1
is a reducing agent.
2
reacts with to form .
3
solution of in contains and ions.
4
The reaction of with hot dilute nitric acid to give is a redox reaction.
43
PYQ 2020
medium
chemistry ID: jee-adva
With respect to hypochlorite, chlorate and perchlorate ions, choose the correct statement(s)
1
The hypochlorite ion is the strongest conjugate base
2
The molecular shape of only chlorate ion is influenced by the lone pair of electrons of
3
The hypochlorite and chlorate ions disproportionate to give rise to identical set of ions
4
The hypochlorite ion oxidizes the sulfite ion
44
PYQ 2021
medium
chemistry ID: jee-adva
Ozonolysis of produces oxide of chlorine. The average oxidation state of chlorine in this oxide is ____.
45
PYQ 2021
medium
chemistry ID: jee-adva
The correct statement(s) related to oxoacids of phosphorous is(are)
1
Upon heating, undergoes a disproportionation reaction to produce and .
2
While can act as a reducing agent, cannot.
3
is a monobasic acid.
4
The H atom of the P-H bond in is not ionizable in water.
46
PYQ 2022
hard
chemistry ID: jee-adva
The reaction of with gives a paramagnetic gas, which upon reaction with produces
1
2
3
4
47
PYQ 2022
medium
chemistry ID: jee-adva
The compound(s) which react(s) with to give boron nitride (BN) is(are)
1
2
3
4
48
PYQ 2022
medium
chemistry ID: jee-adva
Dissolving of white phosphorous in boiling solution in an inert atmosphere gives a gas The amount of (in g) required to completely consume the gas is ___ [Given : Atomic mass of ]
49
PYQ 2022
medium
chemistry ID: jee-adva
Dissolving of white phosphorous in boiling solution in an inert atmosphere gives a gas The amount of (in g) required to completely consume the gas is ___ [Given : Atomic mass of ]
50
PYQ 2022
medium
chemistry ID: jee-adva
The reaction of HClO3 with HCl gives a paramagnetic gas, which upon reaction with O3 produces
1
2
3
4
51
PYQ 2022
medium
chemistry ID: jee-adva
The reaction of HClO3 with HCl gives a paramagnetic gas, which upon reaction with O3 produces
1
2
3
4
52
PYQ 2022
medium
chemistry ID: jee-adva
The reaction of HClO3 with HCl gives a paramagnetic gas, which upon reaction with O3 produces
1
2
3
4
53
PYQ 2022
easy
chemistry ID: jee-adva
The reaction and in water produces a precipitate that dissolves upon the addition of of appropriate concentration The dissolution of the precipitate is due to the formation of
1
2
3
4
54
PYQ 2022
medium
chemistry ID: jee-adva
The reaction of and gives a Xe compound . The number of moles of HF produced by the complete hydrolysis of of is ______
55
PYQ 2022
medium
chemistry ID: jee-adva
The compound(s) which react(s) with to give boron nitride (BN) is(are)
1
2
3
4
56
PYQ 2022
medium
chemistry ID: jee-adva
The reaction and in water produces a precipitate that dissolves upon the addition of of appropriate concentration. The dissolution of the precipitate is due to the formation of
1
2
3
4
 undefined
undefined
