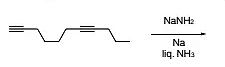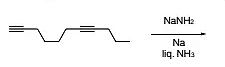Step 1: Understanding the Birch Reduction Process
The Birch reduction is a process in which alkynes react with alkali metals (such as sodium, Na) in liquid ammonia (NH3) to produce trans-alkenes.
This reduction specifically targets internal alkynes, causing them to form trans-alkenes. Terminal alkynes, however, do not undergo this reduction and remain unaffected by the reaction.
Step 2: The Reaction Mechanism
When an alkyne undergoes Birch reduction, the alkali metal (sodium) donates electrons to the alkyne, resulting in the formation of a radical anion. This species reacts with ammonia to form a trans-alkene.
This reduction is selective and works primarily for internal alkynes, converting them to trans-alkenes. Terminal alkynes do not participate in this reaction and remain unchanged.
Step 3: Key Feature of the Reaction
The key characteristic of Birch reduction is the formation of **trans-alkenes** from internal alkynes. This transformation does not occur with terminal alkynes, making them unreactive under these conditions.
Step 4: Conclusion
Option (B) correctly describes this characteristic behavior of alkynes under Birch reduction conditions. It specifies that alkynes undergo Birch reduction in liquid ammonia to form trans-alkenes, while terminal alkynes do not undergo this reduction.

It is a case of Birch reduction. Alkynes on reaction with alkali metal in liq. NH3 gives trans-alkene. But terminal alkynes do not get reduced.
The correct answer is option (B):