JEE-ADVANCED SERIES Chemistry
Law Of Chemical Equilibrium And Equilibrium Constant
11 previous year questions.
Volume: 11 Ques
Yield: Medium
High-Yield Trend
2
2025 1
2024 2
2023 3
2021 2
2020 1
1982 Chapter Questions 11 MCQs
01
PYQ 1982
medium
chemistry ID: jee-adva
The precipitate of C is obtained, when equal volumes of which of the following are mixed?
1
2
3
4
02
PYQ 2020
hard
chemistry ID: jee-adva
Consider the reaction at At time ' ', the temperature of the system was increased to and the system was allowed to reach equilibrium. Throughout this experiment the partial pressure of was maintained at bar. Given below is the plot of the partial pressure of with time. What is the ratio of the standard Gibbs energy of the reaction at to that at ?


03
PYQ 2020
medium
chemistry ID: jee-adva
An acidified solution of is saturated with . What is the minimum molar concentration of required to prevent the precipitation of ?
Use and overall dissociation constant of
Use and overall dissociation constant of
04
PYQ 2021
medium
chemistry ID: jee-adva
For the reaction, , the plot of versus is given below (in solid line), where is the pressure (in bar) of the gas at temperature and bar
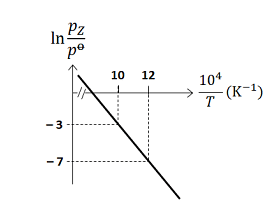
(Given, , where the equilibrium constant, and the gas constant, )
The value of standard enthalpy, (in ) for the given reaction is ______
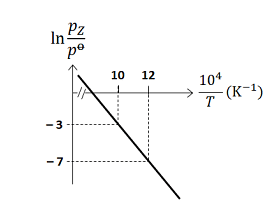
(Given, , where the equilibrium constant, and the gas constant, )
The value of standard enthalpy, (in ) for the given reaction is ______
05
PYQ 2021
hard
chemistry ID: jee-adva
For the reaction, , the plot of versus is given below (in solid line), where is the pressure (in bar) of the gas at temperature and bar
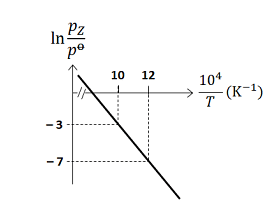
(Given, , where the equilibrium constant, and the gas constant, )
The value of (in ) for the given reaction, at is ______
(Given, , where the equilibrium constant, and the gas constant, )
The value of (in ) for the given reaction, at is ______
06
PYQ 2021
easy
chemistry ID: jee-adva
For the reaction, , the plot of versus is given below (in solid line), where is the pressure (in bar) of the gas at temperature and bar
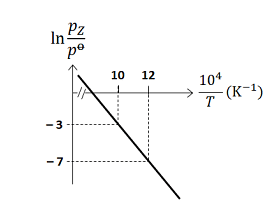
(Given, , where the equilibrium constant, and the gas constant, )
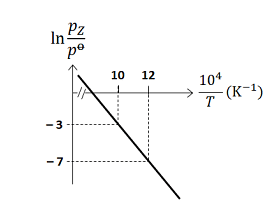
(Given, , where the equilibrium constant, and the gas constant, )
07
PYQ 2023
medium
chemistry ID: jee-adva
In a one-litre flask, 6 moles of A undergoes the reaction A (g) ⇌ P (g). The progress of product formation at two temperatures (in Kelvin), T1 and T2, is shown in the figure:

If T1 = 2T2 and (∆G2Θ − ∆G1Θ) = RT2 ln x, then the value of x is ___ . [∆G1Θ and ∆G2Θ are standard Gibb’s free energy change for the reaction at temperatures T1 and T2, respectively.]
08
PYQ 2023
medium
chemistry ID: jee-adva
In a one-litre flask, 6 moles of A undergoes the reaction A (g) ⇌ P (g). The progress of product formation at two temperatures (in Kelvin), T1 and T2, is shown in the figure:

If T1 = 2T2 and (∆G2Θ − ∆G1Θ) = RT2 ln x, then the value of x is ___ .
[∆G1Θ and ∆G2Θ are standard Gibb’s free energy change for the reaction at temperatures T1 and T2, respectively.]

If T1 = 2T2 and (∆G2Θ − ∆G1Θ) = RT2 ln x, then the value of x is ___ .
[∆G1Θ and ∆G2Θ are standard Gibb’s free energy change for the reaction at temperatures T1 and T2, respectively.]
09
PYQ 2024
easy
chemistry ID: jee-adva
When potassium iodide is added to an aqueous solution of potassium ferricyanide, a reversible reaction is observed in which a complex P is formed. In a strong acidic medium, the equilibrium shifts completely towards P. Addition of zinc chloride to P in a slightly acidic medium results in a sparingly soluble complex Q.
10
PYQ 2025
medium
chemistry ID: jee-adva
The solubility of barium iodate in an aqueous solution prepared by mixing 200 mL of 0.010 M barium nitrate with 100 mL of 0.10 M sodium iodate is . \text{ The value of } \text{ is ------.}
Use: Solubility product constant of barium iodate =
Use: Solubility product constant of barium iodate =
11
PYQ 2025
hard
chemistry ID: jee-adva
The solubility of barium iodate in an aqueous solution prepared by mixing 200 mL of 0.010 M barium nitrate with 100 mL of 0.10 M sodium iodate is . The value of is ------.
Use: Solubility product constant of barium iodate =