Chemical Bonding And Molecular Structure
95 previous year questions.
High-Yield Trend
Chapter Questions 95 MCQs
Correct order of dissociation energy of and is:
Bond energy of
Electronegativity of
is more oxidising than
Electron affinity of
Which of the following has p – d bonding
NO3-
SO3-2
BO3-3
CO3-2
Two
Six
Four
Three
H2O2
O3
O2
O2
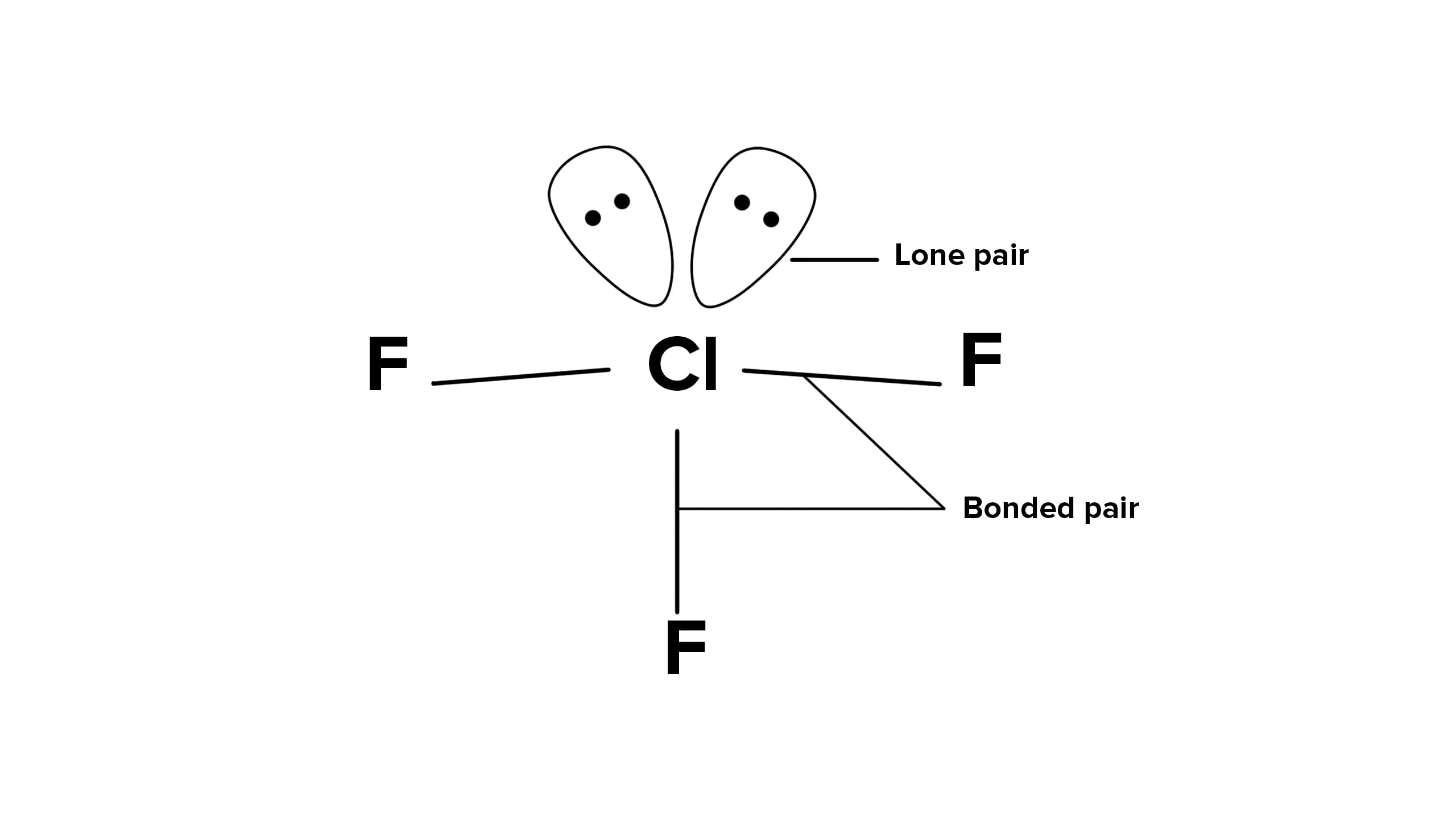
IF3
PCl3
NH3
BF3
BF3
SiF4
SF4
XeF4
ClF3
BF3
AlF3
NF3
NO+
CO
O2-
CN-
BF3 is planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are
sp2 and 8
sp3 and 4
sp3 and 6
sp2 and 6
Which of the following molecules is non-polar in nature
NO2
POCl3
CH2O
SbCl5
Match List - I with List- II. Choose the correct answer from the options given below:
| List - I (Hydrides) | List - II (Nature) | ||
| (a) | MgH2 | (i) | Electron precise |
| (b) | GrH4 | (ii) | Electron deficient |
| (c) | B2H6 | (iii) | Electron rich |
| (d) | HF | (iv) | Ionic |
2
4
1
3
The correct order of energies of molecular orbitals of N2 molecule, is
σ1s<σ*1s<σ2s<σ*2s<σ2pz<(π2px =π2py)<(π*2px=π*2py)<σ*2pz
σ1s<σ*1s<σ2s<σ*2s<σ2pz<σ*2pz<(π2px=π2py)<(π*2px=π*2py)
σ1s<σ*1s<σ2s<σ*2s<(π2px=π2py)<(π*2px=π*2py)<σ2pz<σ*2pz
σ1s<σ*1s<σ2s<σ*2s<(π2px=π2py)<σ2pz<(π*2px=π*2py)<σ*2pz
i)

ii)

iii)

iv)

v)

vi)

vii)

The number of compounds/species which obey Huckel's rule is _____
5
4
6
2
| List - IMolecule | List - IIBond enthalpy (kJ mol-1) |
|---|---|
| (A) HCl | (I) 435.8 |
| (B) N2 | (II) 498 |
| (C) H2 | (III) 946.0 |
| (D) O2 | (IV) 431.0 |
Statement I: A hypothetical diatomic molecule with bond order zero is quite stable.
Statement II: As bond order increases, the bond length increases.
In the light of the above statements, choose the most appropriate answer from the options given below:
Identify the correct orders against the property mentioned:
A. H O NH CHCl - dipole moment
B. XeF XeO XeF - number of lone pairs on central atom
C. O–H C–H N–O - bond length
D. N >O >H - bond enthalpy
Choose the correct answer from the options given below: