Chemical Reactions
30 previous year questions.
High-Yield Trend
Chapter Questions 30 MCQs
3A → 2B,rate of reaction +d[B]/dt is equals to
–
–
H2O involves hydrogen bonding whereas BeF2 is a discrete molecule
H2O is linear and BeF2 is angular
H2O is angular and BeF2 is linear
The electronegativity of F is greater than that of O
It has a very high cryoscopic constant
It is volatile
It is solvent for organic substances
It is readily available


The product ‘C’ is
a)
Select the correct option from the following:








Which of the following reactions is the metal displacement reaction? Choose the right option.
Noble gases have large positive values of electron gain enthalpy
Noble gases are sparingly soluble in water
Noble gases have very high melting and boiling points
Noble gases have weak dispersion forces
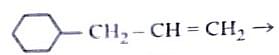
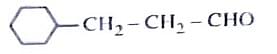
(ii) CrO3
(ii)
(iii) PCC
(ii)
(iii) alk. KMnO4
(i) H2O/H+
(ii) PCC
A. Tollen's reagent
B. Schiff's reagent
C. HCN
D NH2OH
E. NaHSO3
Choose the correct options from the given below:
Identify the major product C formed in the following reaction sequence: 
Given R=8.314 J K-1 mol-1, log 4=0.6021
3ROH+PCI3→3RCI+A
ROH+PCI5→RCI+HCI+B
H3PO3 and POCI3
List-I | List-II | ||
| (A) |  | (I) |  |
| (B) |  | (II) | CrO3 |
| (C) |  | (III) | KMnO4/KOH, |
| (D) |  | (IV) | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:

‘P’ is






