KCET SERIES Chemistry
Electrochemistry
33 previous year questions.
Volume: 33 Ques
Yield: High
High-Yield Trend
5
2026 1
2023 5
2022 3
2021 2
2019 2
2018 1
2017 1
2016 3
2015 3
2014 1
2009 1
2008 2
2006 1
2005 1
2001 Chapter Questions 33 MCQs
01
PYQ 1992
medium
chemistry ID: kcet-199
Which of the following is not a strong electrolyte
1
2
3
4
02
PYQ 2001
medium
chemistry ID: kcet-200
The quantity of electricity required to liberate of hydrogen at STP from acidified water is
1
965 C
2
4825 C
3
9650 C
4
96500 C
03
PYQ 2005
medium
chemistry ID: kcet-200
The standard electrode potential is measured by
1
Electrometer
2
Voltmeter
3
Pyrometer
4
Galvanometer
04
PYQ 2006
medium
chemistry ID: kcet-200
When a quantity of electricity is passed through solution, of Copper gets deposited. If the same quantity of electricity is passed through acidulated water, then the volume of liberated at will be [Given ]
1
2
3
4
05
PYQ 2006
medium
chemistry ID: kcet-200
Which of the following does not conduct electricity ?
1
fused NaCl
2
solid NaCl
3
brine solution
4
Copper
06
PYQ 2008
easy
chemistry ID: kcet-200
Which of the following electrolytic solutions has the least specific conductance ?
1
2
3
4
07
PYQ 2009
easy
chemistry ID: kcet-200
In the electrolysis of acidulated water, it is desired to obtain of Hydrogen per second under condition. The current to be passed is
1
2
3
4
08
PYQ 2014
medium
chemistry ID: kcet-201
For = milli mole milli mole, is .The pH of the solution is nearly equal to
1
3
2
4
3
2
4
5
09
PYQ 2014
medium
chemistry ID: kcet-201
For hydrogen - oxygen fuel cell at one and for the cell is approximately, (Given )
1
2
3
4
10
PYQ 2014
medium
chemistry ID: kcet-201
Impure copper containing as impurities is electrolytically refined. A current of for decreased the mass of the anode by and increased the mass of cathode by . Percentage of iron in impure copper is (Given molar mass , molar mass )
1
2
3
4
11
PYQ 2015
medium
chemistry ID: kcet-201
In fuel cell the reaction occurring at cathode is
1
2
3
4
12
PYQ 2015
medium
chemistry ID: kcet-201
How many Coulombs of electricity are required for the oxidation of one mol of water to dioxygen ?
1
2
3
4
13
PYQ 2015
easy
chemistry ID: kcet-201
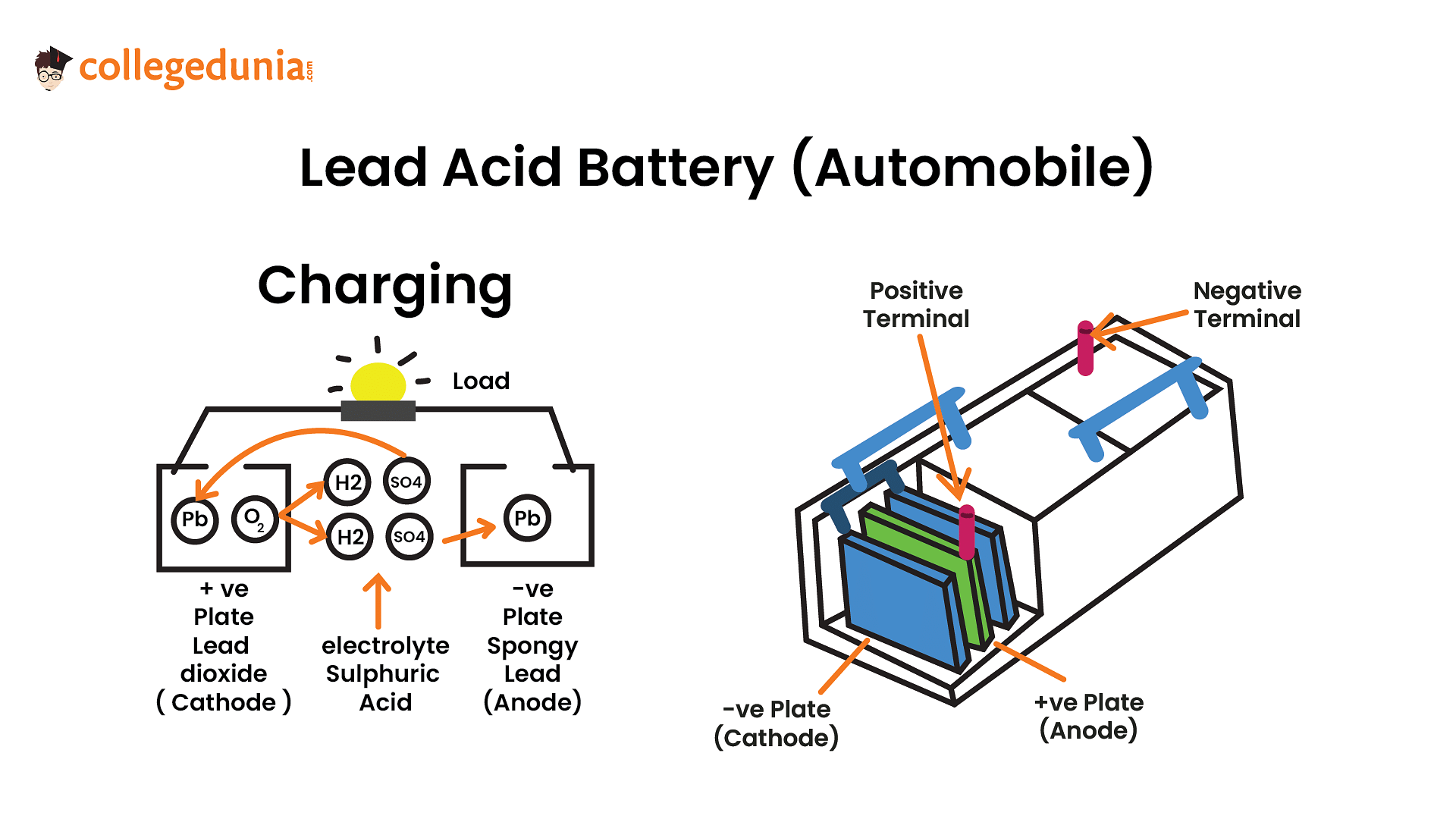
While charging the lead storage battery
1
on anode is reduced to
2
on cathode is reduced to
3
on cathode is oxidized to
4
on anode is oxidized to
14
PYQ 2016
medium
chemistry ID: kcet-201
Which of the following is incorrect in a galvanic cell ?
1
Oxidation occurs at anode.
2
Reduction occurs at cathode.
3
The electrode at which electrons are gained is called cathode.
4
The electrode at which electrons are lost is called cathode
15
PYQ 2017
medium
chemistry ID: kcet-201
By passing electric current, is converted into according to the following equation How many moles of will be formed when three Faradays of charge is passed through ?
1
2
3
4
16
PYQ 2018
medium
chemistry ID: kcet-201
At a particular temperature, the ratio of molar conductance to specific conductance of 0.01 M NaCl solution is
1
2
3
4
17
PYQ 2018
medium
chemistry ID: kcet-201
For a cell reaction involving two electron changes, at . The equilibrium constant of the reaction is
1
2
3
10
4
18
PYQ 2019
medium
chemistry ID: kcet-201
Give: E0Mn+7|Mn+2 = 1.5 V and E0Mn+4|Mn+2 = 1.2 V, then E0Mn+7|Mn+4 is
1
1.7 V
2
2.1 V
3
0.3 V
4
0.1 V
19
PYQ 2019
medium
chemistry ID: kcet-201
An aqueous solution of CuSO4 is subjected to electrolysis using inert electrodes. The pH of the solution will
1
decrease
2
increase or decrease depending on the strength of the current.
3
increase
4
remains unchanged
20
PYQ 2021
medium
chemistry ID: kcet-202
at 25° C for the cell is 0.22 V. The equilibrium constant at 25° C is
1
2.8 × 107
2
5.2 × 108
3
2.8 × 105
4
5.2 × 104
21
PYQ 2021
medium
chemistry ID: kcet-202
The resistance of 0.01 m KCl solution at 298 K is 1500Ω. If the conductivity of 0.01 m KCl solution at 298 K is 0.1466 × 10-3 S cm-1. The cell constant of the conductivity cell in cm-1 is
1
0.219
2
0.291
3
0.301
4
0.194
22
PYQ 2021
medium
chemistry ID: kcet-202
Consider the following electrodes
P = Zn2+ (0.0001 M)/Zn Q = Zn2+ (0.1 M)/Zn
R = Zn2+ (0.01 M)/Zn S = Zn2+ (0.001 M)/Zn
E°Zn/Zn2+ = 0.76 V Electrode potentials of the above electrodes in volts are in the order
P = Zn2+ (0.0001 M)/Zn Q = Zn2+ (0.1 M)/Zn
R = Zn2+ (0.01 M)/Zn S = Zn2+ (0.001 M)/Zn
E°Zn/Zn2+ = 0.76 V Electrode potentials of the above electrodes in volts are in the order
1
P > S > R > Q
2
S > R > Q > P
3
Q > R > S > P
4
P > Q > R > S
23
PYQ 2022
easy
chemistry ID: kcet-202
A nucleic acid whether DNA or RNA on complete hydrolysis, two purine bases, two pyrimidine bases, a pentose sugar and phosphoric acid.Nucleotides which are intermediate products in the hydrolysis contain
1
Purine or pyrimidine base and ortho phosphoric acid
2
Purine or pyrimidine base and pentose sugar
3
Purine or pyrimidine base, a pentose sugar and ortho phosphoric acid
4
a purine base, a pentose sugar and ortho phosphoric acid
24
PYQ 2022
medium
chemistry ID: kcet-202
For spontaneity of a cell, which is correct ?
1
ΔG = +ve, ΔE = +ve
2
ΔG = 0, ΔE = 0
3
ΔG = -ve
4
ΔG = -ve, ΔE = 0
25
PYQ 2022
medium
chemistry ID: kcet-202
Specific Conductance of 0.1 M HNO3 is 6.3 x 10-2 ohm -1 cm-1. The molar conductance of the solution is
1
6300 ohm-1 cm2 mol-1
2
630 ohm-1 cm2 mol-1
3
63.0 ohm-1 cm2 mol-1
4
315 ohm-1 cm2 mol-1
26
PYQ 2022
medium
chemistry ID: kcet-202
The molar conductivity is maximum for the solution of concentration
1
0.005 M
2
0.004 M
3
0.001 M
4
0.002
27
PYQ 2022
medium
chemistry ID: kcet-202
In fuel cell ___ are used as catalysts.
1
Zinc - Mercury
2
Platinum - Palladium
3
Lead - Manganese
4
Nickel - Cadmium
28
PYQ 2023
medium
chemistry ID: kcet-202
The resistance of 0.1 M weak acid HA in a conductivity cell is 2 × 103 Ohm. The cell constant of the cell is 0.78 C m-1 and of acid HA is 390 S cm2 mol-1. The pH of the solution is
1
3.3
2
4.2
3
5
4
3
29
PYQ 2026
medium
chemistry ID: kcet-202
During the electrolysis of acidified water, 16 g of O gas is formed at anode. The volume of H gas liberated at cathode under STP conditions is
1
22.4 L
2
11.2 L
3
2.24 L
4
1.12 L
30
PYQ 2026
easy
chemistry ID: kcet-202
The conductivity of centimolar solution of KCl at 298 K is 0.021 cm . The resistance of the cell containing the solution at 298 K is 60 . The value of cell constant is
1
3.28 cm
2
1.26 cm
3
3.54 cm
4
1.34 cm
31
PYQ 2026
hard
chemistry ID: kcet-202
is equal to ___________
1
2
3
4
32
PYQ 2026
easy
chemistry ID: kcet-202
During the electrolysis of acidified water, 16 g of O gas is formed. The volume of H gas liberated at cathode under STP conditions is
1
22.4 L
2
11.2 L
3
2.24 L
4
1.12 L
33
PYQ 2026
medium
chemistry ID: kcet-202
Given below are the half-cell reactions:
(E = -1.18 V)
(E = +1.51 V)
The E for will be \textunderscore\textunderscore\textunderscore\textunderscore
(E = -1.18 V)
(E = +1.51 V)
The E for will be \textunderscore\textunderscore\textunderscore\textunderscore
1
- 2.69 V, the reaction will not occur (Non-Spontaneous)
2
- 2.69 V, the reaction will occur (Spontaneous)
3
- 0.33 V, the reaction will not occur (Non-Spontaneous)
4
- 0.33 V, the reaction will occur (Spontaneous)