KCET SERIES Chemistry
The Valence Shell Electron Pair Repulsion Vsepr Theory
3 previous year questions.
Volume: 3 Ques
Yield: Medium
High-Yield Trend
1
2020 1
2019 1
2016 Chapter Questions 3 MCQs
01
PYQ 2016
medium
chemistry ID: kcet-201
The shape of is,
1
Square planar
2
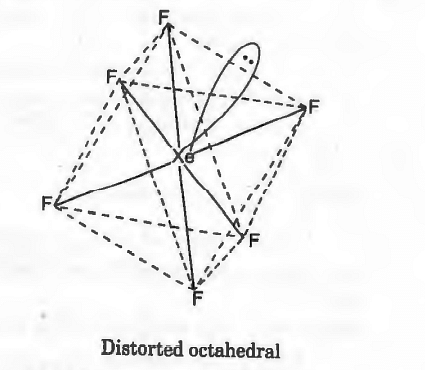
Distorted octahedral
3
Square pyrimidal
4
Pyramidal
02
PYQ 2019
medium
chemistry ID: kcet-201
According to the VSEPR theory, the shape of would be
1
Linear
2
Triangular planar
3
Pyramidal
4
Square planar
03
PYQ 2020
medium
chemistry ID: kcet-202
Incorrectly matched pair is:
1
- pyramidal
2
- tetrahedral
3
- disorted octahedral
4
- square pyramidal