Specific Heat Capacity
14 previous year questions.
High-Yield Trend
Chapter Questions 14 MCQs




Given below are two statements:
Statement-I : Susceptibilities of paramagnetic and ferromagnetic substances increase with decrease in temperature.
Statement-II : Diamagnetism is a result of orbital motions of electrons developing magnetic moments opposite to the applied magnetic field.
Choose the correct answer from the options given below:-
A copper block of mass 5.0 kg is heated to a temperature of 500°C and is placed on a large ice block. What is the maximum amount of ice that can melt?
[Specific heat of copper 0.39 J g–1 °C–1 and latent heat of fusion of water : 335 J g–1]
(Given : specific heat capacity of water : )
(Assume that the given diatomic gas has no vibrational mode).
Let be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, is :
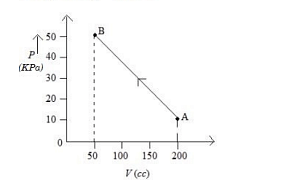
The pressure of a gas changes linearly with volume from to as shown in figure If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be Is
A water heater of power is used to heat water. The specific heat capacity of water is .The efficiency of heater is .Time required to heat of water from to is___
(Assume that the specific heat capacity of water remains constant over the temperature range of the water)
An engine operating between the boiling and freezing points of water will have
A. efficiency more than 27%
B. efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
C. efficiency equal to 27%
D. efficiency less than 27%
Match List-I with List-II.
| List-I | List-II |
| (A) Heat capacity of body | (I) |
| (B) Specific heat capacity of body | (II) |
| (C) Latent heat | (III) |
| (D) Thermal conductivity | (IV) |
(A)-(II), (B)-(III), (C)-(I), (D)-(IV)