Thermodynamics
174 previous year questions.
High-Yield Trend
Chapter Questions 174 MCQs




In the reported figure, there is a cyclic process ABCDA on a sample of 1 mol of a diatomic gas. The temperature of the gas during the process A B and C D are and ( ) respectively. Choose the correct option out of the following for work done if processes BC and DA are adiabatic. 
[Given 1 cal = 4.2 J and specific heat of water = 1 cal g C ]
Thermodynamic process is shown below on a P-V diagram for one mole of an ideal gas. If then the ratio of temperature is: The process is = constant. 
The P-V diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process CD is (use γ = 1.4) : 
A perfect gas undergoes a cyclic process ABCA. A→B: Isothermal expansion ( ). B→C: Isobaric compression to . C→A: Isochoric change to . Total work done is :
List I: (a) Isothermal (b) Isochoric (c) Adiabatic (d) Isobaric.
List II: (i) Pressure constant (ii) Temperature constant (iii) Volume constant (iv) Heat content constant.
[R = 8.314 J mol K ]
If one mole of an ideal gas at is allowed to expand reversibly and isothermally (A to B) its pressure is reduced to one-half of the original pressure. This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value (B → C). Then it is restored to its initial state by a reversible adiabatic compression (C to A). The net work done by the gas is equal to : 
Given below are two statements
Statement-I: When μ amount of an ideal gas undergoes adiabatic change from state (P1, V1, T1) to state (P2, V2, T2), then work done is
,where
and R = universal gas constant.
Statement-II: In the above case, when work is done on the gas, the temperature of the gas would rise.
Choose the correct answer from the options given below.
8
4
A sample of an ideal gas is taken through the cyclic process ABCA as shown in figure. It absorbs, 40 J of heat during the part AB, no heat during BC and rejects 60 J of heat during CA. A work of 50 J is done on the gas during the part BC. The internal energy of the gas at A is 1560 J. The work done by the gas during the part CA is:

The total internal energy of two mole monoatomic ideal gas at temperature T=300 K will be _____J.
(Given R = 8.31 J/mol.K)
(Given )
In a carnot engine, the temperature of reservoir is 527°C and that of sink is 200 K. If the work done by the engine when it transfers heat from reservoir to sink is 12000 kJ, the quantity of heat absorbed by the engine from reservoir is _____ J.
Assertion A: Efficiency of a reversible heat engine will be highest at temperature of cold reservoir
Reason R: The efficiency of Carnot's engine depends not only on temperature of cold reservoir but it depends on the temperature of hot reservoir too and is given as
In the light of the above statements, choose the correct answer from the options given below
Ratio of molar heat capacity at constant pressure and at constant volume for monoatomic and diatomic gas is?
25 : 21
21 : 25
16 : 25
25 : 16




Statement I: If heat is added to a system, its temperature must increase.
Statement II: If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
A. Final temperature of system will be 0°C.
B. Final temperature of the system will be greater than 0°C.
C. The final system will have a mixture of ice and water in the ratio of 5:1.
D. The final system will have a mixture of ice and water in the ratio of 1:5.
E. The final system will have water only.
Choose the correct answer from the options given below:





Given below are two statements:
Statement 1: If a capillary tube is immersed first in cold water and then in hot water, the height of capillary rise will be smaller in hot water.
Statement 2: If a capillary tube is immersed first in cold water and then in hot water, the height of capillary rise will be smaller in cold water.
In the light of the above statements, choose the most appropriate option from the options given below:




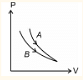
For process A : PV = constant For process B : PV = constant

Match List-I with List-II.
Choose the correct answer from the options given below :
A piston of mass M is hung from a massless spring whose restoring force law goes as F = -kx, where k is the spring constant of appropriate dimension. The piston separates the vertical chamber into two parts, where the bottom part is filled with 'n' moles of an ideal gas. An external work is done on the gas isothermally (at a constant temperature T) with the help of a heating filament (with negligible volume) mounted in lower part of the chamber, so that the piston goes up from a height to , the total energy delivered by the filament is (Assume spring to be in its natural length before heating) 
An ideal gas has undergone through the cyclic process as shown in the figure. Work done by the gas in the entire cycle is _____ J. (Take ) 
Match the List-I with List-II.
Choose the correct answer from the options given below:
The internal energy of air in sized room at 1 atmospheric pressure will be . (Consider air as a diatomic molecule)
A. The work done by gas during the process is zero.
B. The heat added to the gas is different from the change in its internal energy.
C. The volume of the gas is increased.
D. The internal energy of the gas is increased.
E. The process is isochoric (constant volume process).
Choose the correct answer from the options given below:
An amount of ice of mass kg and temperature is transformed to vapor of temperature by applying heat. The total amount of work required for this conversion is,
(Take, specific heat of ice = 2100 J kg K ,
specific heat of water = 4180 J kg K ,
specific heat of steam = 1920 J kg K ,
Latent heat of ice = J kg ,
Latent heat of steam = J kg )
Assertion (A): With the increase in the pressure of an ideal gas, the volume falls off more rapidly in an isothermal process in comparison to the adiabatic process.
Reason (R): In an isothermal process, , while in an adiabatic process . Here, is the ratio of specific heats, is the pressure and is the volume of the ideal gas.
In the light of the above statements, choose the correct answer from the options given below:

Match List-I with List-II.
is the specific heat ratio of monoatomic gas A having 3 translational degrees of freedom. is the specific heat ratio of polyatomic gas B having 3 translational, 3 rotational degrees of freedom and 1 vibrational mode. If then the value of is ___.
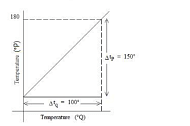
Which of the following best represents the temperature versus heat supplied graph for water, in the range of to ? 


Find heat given to gas to take it from A to B. (Given: S.I. units, Pa, Pa, S.I. units, moles) 

Assertion (A): The individual atoms in a ferromagnetic material possess a magnetic dipole moment and interact with one another in such a way that they spontaneously align themselves forming domains.
Reason (R): At high enough temperature, the domain structure of ferromagnetic material disintegrates. Thus, magnetization will disappear at high enough temperature known as Curie temperature.
In the light of the above statements, choose the correct answer from the options given below :
In the following diagram, the equation of state along the curved path is given by where is a constant. The total work done in the closed path is: 
For a gas P-V curve is given as shown in the diagram. Curve path follows equations . Find work done by gas in given cyclic process. 


Two balls made of the same material collide perfectly inelastically as shown. The energy lost in the collision is completely utilized in raising the temperature of each ball. Find the rise in temperature of the balls. (Specific heat cal/kg- C): 
Two rods of equal length each are joined together end to end. The coefficients of linear expansion of the rods are and . Their initial temperature is , which is increased to . Find the final length of the combination (in cm).
Given:
,
The change in internal energy of the gas is:


