Chemical Bonding And Molecular Structure
158 previous year questions.
High-Yield Trend
Chapter Questions 158 MCQs
The number of hydrogen bonded water molecule(s) associated with stoichiometry is ____________
Match List - I with List - II : 
Choose the most appropriate answer from the options given below :
The compound/s which will show significant intermolecular H-bonding is/are : 
Assertion A : Lithium halides are some what covalent in nature.
Reason R : Lithium possess high polarisation capability.
In the light of the above statements, choose the most appropriate answer from the options given below :
(A) SO
(B) NO
(C) PCl
(D) CO
(Round off to the Nearest Integer).
List-I (Compound) List-II (Shape)
A I bent
B II square pyramidal
C III trigonal bipyramidal
D IV octahedral
Choose the correct answer from the optionsgiven below :
In which of the following pairs, electron gain enthalpies of constituent elements are nearly the same or identical ?
(A) and
(B) and
(C) and
(D) and
Choose the correct answer from the options given below :
A.
B.
C.
D.
N2,N2+,N2−,N22−,O2,O2+,O2−,O22−
the number of species showing diamagnetism is ________.
(A) NO
(B) N2
(C) O2
(D) C2
(E) B2
Choose the most appropriate answer from the options given below :
Br– and Be2+
and
Amongst these oxoacids, the number of those with peroxo (O–O) bonds is_____.
Statement I: The compound

(A) is optically active.
Statement II:

is a mirror image of the above compound A.
In the light of the above statement, choose the most appropriate answer from the options given below.
Consider the species
Choose the correct option with respect to these species.
In the given reaction,

The number of π electrons present in the product ‘P’ is_____.
Match List I with List II.
| List-I | List-II | ||
|---|---|---|---|
| A | Sulphate | I | Pesticide |
| B | Fluoride | II | Bending of bones |
| C | Nicotine | III | Laxative effect |
| D | Sodium arsinite | IV | Herbicide |
Choose the correct answer from the options given below:
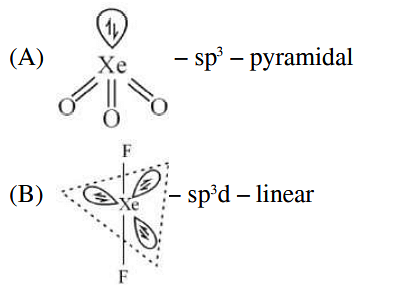
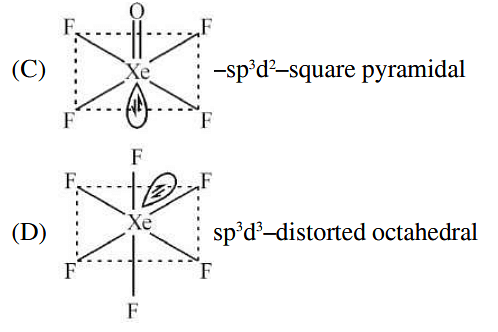
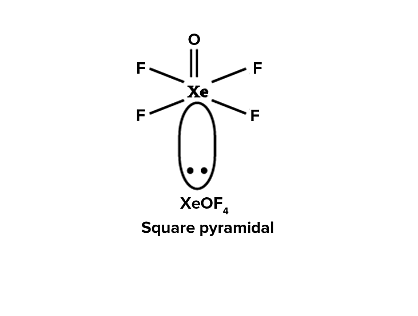
Match List I with List II :
| List-I (molecule) | List-II (hybridization; shape) | ||
|---|---|---|---|
| A | XeO3 | i | sp3d ; linear |
| B | XeF2 | ii | sp3; pyramidal |
| C | XeOF4 | iii | sp3d3; distorted octahedral |
| D | XeF6 | iv | sp3d2 ;square pyramidal |
Choose the correct answer from the options given below:
Number of unpaired electron in highest occupied molecular orbital of following species is : ?
0, 1, 2, 1
2, 1, 2, 1
0, 1, 0, 1
2, 1, 0, 1
Statement II : The crossed arrow of the dipole moment symbolizes the direction of the shift of charges in the molecules.
In the light of the above statements, choose the most appropriate answer from the options given below:
- Polarising power of cation
- Polarisability of the anion
- Distortion caused by cation
- Polarisability of cation
A.
B.
C.
D.
E.
Choose the correct answer from the options given below:
| Column I | Column II | ||
| (A). | ClO2- | (1). | Linear |
| (B). | N3- | (2). | Tetrahedral |
| (C). | NH4+ | (3). | Bent |
| (D). | SF4 | (4). | See-Saw |
Which of the following has maximum number of l.p. at central atom?
ClO3-
SF4
XeF4
I3-
What is the correct order of acidity of the protons marked A-D in the given compounds?
HB > HA > HD > HC
HA > HB > HC > HD
| List I | Species | List II | Geometry/ Shape |
| A | H3 O+ | I | Tetrahedral |
| B | Acetylide anion | II | Linera |
| C | NH4 + | III | Pyramidal |
| D | ClO2– | IV | Bent |
Choose correct answer from the options given below:
For molecule consider the following:
[(A)] Number of lone pairs on oxygen is 2.
[(B)] F--O--F angle is less than .
[(C)] Oxidation state of O is .
[(D)] Molecule is bent ‘V’-shaped.
[(E)] Molecular geometry is linear.
Correct options are:
A, B, D only
A, C, D only
C, D, E only
B, E, A only
ClO3– , XeF4, SF4 and I3–
N ClF SO K O LiF
N SO ClF K O LiF

Choose the correct answer from the options given below:
Statement (I) : Both metal and non-metal exist in p and d-block elements.
Statement (II) : Non-metals have higher ionisation enthalpy and higher electronegativity than the metals.
In the light of the above statements, choose the most appropriate answer from the option given below:
Resonance effect
Inductive effect
Electromeric effect
,
The number of species from the following which are paramagnetic and with bond order equal to one is ___.
Species: H2, He2+, O2-, N2, O22-, F2, Ne2+, B2
Species: H2, He2+, O2-, N2, O22-, F2, Ne2+, B2
The number of molecules/ions having trigonal bipyramidal shape is:
A. have the same energy
B. have the minimum overlap
C. have same symmetry about the molecular axis
D. have different symmetry about the molecular axis.
Choose the most appropriate from the options given below:




(Given )
Statement (I): The oxidation state of an element in a particular compound is the charge acquired by its atom on the basis of electron gain enthalpy consideration from other atoms in the molecule.
Statement (II): - bond formation is more prevalent in second period elements over other periods.
In the light of the above statements, choose the {most appropriate answer from the options given below:
| List-I (Molecule) | List-II (Shape) |
|---|---|
| (A) | (I) Square pyramid |
| (B) | (II) Tetrahedral |
| (C) | (III) Trigonal pyramidal |
| (D) | (IV) Trigonal bipyramidal |
| List - I | List - II |
|---|---|
| (A) ICl | (IV) Linear |
| (B) ICl3 | (I) T-Shape |
| (C) ClF5 | (II) Square pyramidal |
| (D) IF7 | (III) Pentagonal bipyramidal |
(Given atomic number: C : 6, Na : 11, O : 8, Fe : 26, Cr : 24)
HF, H , H S, CO , NH , BF , CH , CHCl , SiF , H O, BeF
| List-I (Compound / Species) | List-II (Shape / Geometry) |
|---|---|
| (A) | (I) Tetrahedral |
| (B) | (II) Pyramidal |
| (C) | (III) See saw |
| (D) | (IV) Bent T-shape |
Assertion (A): NH and NF molecule have pyramidal shape with a lone pair of electrons on nitrogen atom. The resultant dipole moment of NH is greater than that of NF .
Reason (R): In NH , the orbital dipole due to lone pair is in the same direction as the resultant dipole moment of the N H bonds. F is the most electronegative element.
In the light of the above statements, choose the correct answer from the options given below:
Statement II: In NH3, the orbital dipole due to the lone pair and the dipole moment of NH bonds are in opposite directions, but in NF3, the orbital dipole due to the lone pair and the dipole moments of N-F bonds are in the same direction.
In light of the above statements, choose the most appropriate from the options given below:
Which of the following molecules(s) show/s paramagnetic behavior?
A and B
B and C
D and B
A and D
Match the LIST-I with LIST-II
| LIST-I | LIST-II |
| A. PF5 | I. dsp2 |
| B. SF6 | II. sp3d |
| C. Ni(CO)4 | III. sp3d2 |
| D. [PtCl4]2- | IV. sp3 |
Choose the correct answer from the options given below:
Given below are two statements:
Statement I : The N-N single bond is weaker and longer than that of P-P single bond
Statement II : Compounds of group 15 elements in +3 oxidation states readily undergo disproportionation reactions.
In the light of above statements, choose the correct answer from the options given below
Match the LIST-I with LIST-II:

Choose the correct answer from the options given below :
Resonance in X Y can be represented as 
The enthalpy of formation of X Y is 80 kJ mol , and the magnitude of resonance energy of X Y is:
Match the LIST-I with LIST-II.
Choose the correct answer from the options given below :
Given below are two statements :
Statement I : Wet cotton clothes made of cellulose based carbohydrate takes comparatively longer time to get dried than wet nylon polymer based clothes.
Statement II : Intermolecular hydrogen bonding with water molecule is more in nylon-based clothes than in the case of cotton clothes.
In the light of the above statements, choose the Correct answer from the options given below
The number of molecules/ions that show linear geometry among the following is _____. SO₂, BeCl₂, CO₂, N₃⁻, NO₂, F₂O, XeF₂, NO₂⁺, I₃⁻, O₃

Choose the correct answer from the options given below:
The formal charges on the atoms marked as (1) to (4) in the Lewis representation of molecule respectively are 
Compound 'P' undergoes the following sequence of reactions : (i) NH₃ (ii) Q (i) KOH, Br₂ (ii) CHCl₃, KOH (alc), NC-CH₃. 'P' is : 

[A.] The reaction proceeds through a more stable radical intermediate.
[B.] The role of peroxide is to generate radical.
[C.] During this reaction, benzene is formed as a byproduct.
[D.] -Bromo- -phenylethane is formed as a minor product.
[E.] The same reaction in absence of peroxide proceeds via a carbocation intermediate. Choose the correct answer.
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are : 
Statement II: A group 15 element's hydride has the lowest boiling point among corresponding hydrides of other group 15 elements. The maximum covalency of that element is 4.
In the light of the above statements, choose the correct answer from the options given below.
Statement I : The correct order in terms of bond dissociation enthalpy is .
Statement II : The correct trend in the covalent character of the metal halides is , and .
In the light of the above statements, choose the correct answer from the options given below :
Select the correct option having formal charge of I, II, III and IV respectively.

Statement I: The number of pairs among [SiO₂, CO₂], [SnO, SnO₂], [PbO, PbO₂] and [GeO, GeO₂], which contain oxides that are both amphoteric is 2.
Statement II: BF₃ is an electron deficient molecule, can act as a Lewis acid, forms adduct with NH₃ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below:
Statement I: HX bond length is higher in HCl than HF.
Statement II: The lowest boiling point in hydride of group 15 element is having covalency 4.

\text{NH}_4^+, \, \text{XeF}_4, \, [\text{NiCl}_4]^{2-}, \, [\text{PtCl}_4]^{2-}, \, [\text{Cu(NH}_3\text{)}_4]^{2+}, \, \text{BF}_3, \, [\text{Ni(CO)}_4]
Statement-II : Among , , and the highest bond order is found in .

[2mm] Statement-II : The melting point order is :