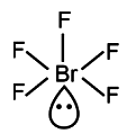
Hybridisation
16 previous year questions.
High-Yield Trend
Chapter Questions 16 MCQs
Amongst the above molecule(s)/ion(s), the number of molecule(s)/ion(s) having hybridisation is _____.
Given below are two statements:
Statement I: [Ni(CN)4]2– is square planar and diamagnetic complex, with dsp2 hybridization for Ni but [Ni(CO)4] is tetrahedral, paramagnetic and with sp3-hybridization for Ni.
Statement II: [NiCl4]2– and [Ni(CO)4] both have same d-electron configuration, have same geometry and are paramagnetic.
In light of the above statements, choose the correct answer from the options given below :
only
only
only
only
and
[Co(NH3)6]3+
SF6
BrF5
[PtCl4]2-
PCI5
BrF5
PF5
[Ni(CN)4]2-
Match List - I with List - II.
| List - I (Complex) | List - II (Hybridisation) |
|---|---|
| (A) | (I) |
| (B) | (II) |
| (C) | (III) |
| (D) | (IV) |
Choose the correct answer from the options given below:
Concentrated nitric acid is labelled as 75% by mass. The volume in mL of the solution which contains 30 g of nitric acid is:
Given: Density of nitric acid solution is 1.25 g/mL.
Arrange the following in increasing order of solubility product:
Match List - I with List - II.
List - I (Saccharides) List - II (Glycosidic linkages found)
(A) Sucrose (I)
(B) Maltose (II) and
(C) Lactose (III)
(D) Amylopectin (IV)
Choose the correct answer from the options given below: