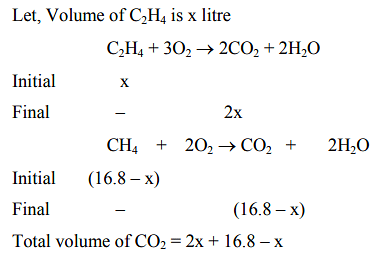Thermodynamics
166 previous year questions.
High-Yield Trend
Chapter Questions 166 MCQs








for KCl = -436.7 kJ ; for K = 89.2 kJ ;
for K = 419.0 kJ ; for = -348.6 kJ ;
for = 243.0 kJ .
The magnitude of lattice enthalpy of KCl in kJ is _________. (Nearest integer)}
[Use: R=8.3 J mol K ]
[ Given : Specific heat of water
Density of water ]
(Assume no volume change on mixing)
Data given for the following reaction is as follows:
FeO(s) + C(graphite) Fe(s) + CO(g)
The minimum temperature in K at which the reaction becomes spontaneous is _________. (Integer answer) 
[Use : H (aq) + OH (aq) H O(l) ; = -57.1 kJ mol
Specific heat of H O = 4.18 J K g
density of H O = 1.0 g cm
Assume no change in volume of solution on mixing.]
For water at 100 C and 1 bar,  (Round off to the Nearest Integer)
(Round off to the Nearest Integer)
[Use : R=8.31 J mol K ]
[Assume volume of H O(l) is much smaller than volume of H O(g). Assume H O(g) can be treated as an ideal gas]
List-I
(A) Spontaneous process
(B) Process with ,
(C)
(D) Exothermic Process
List-II
(I)
(II)
(III) Isothermal and isobaric process
(IV) [Bond energies of molecules in reactants] – [Bond energies of product molecules]
Choose the correct answer from the options given below :
[Assume steam to be an ideal gas. Given ΔvapHΘ for water at 373 K and 1 bar is 41.1 kJ mol–1; R = 8.31 J K–1 mol–1]
The enthalpy change for the conversion of to is (-)_____________ (Nearest integer)
Given : , ,
The enthalpy of ionization of as calculated by the student is _____ (nearest integer)
of ethane undergoes combustion at in a bomb calorimeter The temperature of calorimeter system (including the water) is found to rise by The heat evolved during combustion of ethane at constant pressure is ___ (Nearest integer) [Given : The heat capacity of the calorimeter system is Assume ideal gas behaviour Atomic mass of and are 12 and respectively]
Given
The value of for a reaction A ⇋ B is
(Given,
= –54.67 kJ
= 10 kJ
and R = 8.314 J
2.303 × 8.314 × 298 = 5705)
| LIST I Isomeric pairs | LIST II Type of isomers | ||
| A | Propanamine and N-Methylethanamine | I | Metamers |
| B | Hexan-2-one and Hexan-3-one | II | Positional isomers |
| C | Ethanamide and Hydroxyethanimine | II | Functional isomers |
| D | o-nitrophenol and p-nitrophenol | IV | Tautomers |
The value of log10K for a reaction A ⇋ B is (Given ΔHº298K = –54.67 kJmol–1 ΔSº298K = 10 kJmol–1 and R = 8.314 JK–1mol–1 2.303 × 8.314 × 298 = 5705)
(A)
(B)
(C)
(D)
Choose the most appropriate answer from the options given below:
Volume. Molar heat capacity, Molarity, E\theta cell. Gibbs free energy change. Molar mass, Mole
.
for the reaction is ______ kJ. (Given R= )
Assertion (A): Enthalpy of neutralisation of strong monobasic acid with strong monoacidic base is always –57 kJ mol–1
Reason (R): Enthalpy of neutralisation is the amount of heat liberated when one mole of H+ ions furnished by acid combine with one mole of –OH ions furnished by base to form one mole of water. In the light of the above statements.
Choose the correct answer from the options given below.

Choose the correct answer from options given below:
(Given )
where the bond energies of , , are , , , and respectively, is _________ .

(1) standard Enthalpy of formation of 1 mol of C H (l), for the reaction 6C(graphite) + 3H (g) → C H (l) is 48.5 kJ mol .
(2) Standard Enthalpy of formation of 1 mol of (g), for the reaction 6(graphite) + → CO (g) is -393.5 kJ mol .
(3) Standard Enthalpy of formation of 1 mol of H O(l), for the reaction H (g) + O (g) → H O(l) is -286 kJ mol .
Which of the following is not correct?
(Given Molar mass in g mol−1: C=12.0, H=1.0, O=16.0).
For a given reaction , is related to as given in the table. Given: . Which of the following is true?
| (mol/L) | (min) |
|---|---|
| 0.100 | 200 |
| 0.025 | 100 |
A. The order of the reaction is .
B. If is 1 M, then is min.
C. The order of the reaction changes to 1 if the concentration of reactant changes from 0.100 M to 0.500 M.
D. is 800 min for M.
A, B and D only
A and C only
A and B only
C and D only
Two vessels A and B are connected via stopcock. Vessel A is filled with a gas at a certain pressure. The entire assembly is immersed in water and allowed to come to thermal equilibrium with water. After opening the stopcock the gas from vessel A expands into vessel B and no change in temperature is observed in the thermometer. Which of the following statement is true? 
B.
C.
D.
E.
Mg>Al>C>O
Al>B>N>F
Be>Mg>Al>Si
Si>P>Cl>F
(B) Cellulose
(C) Glycogen
(D) Amylopectin
List - II: (I) -C1-C4 plant
(II) -C1-C4 animal
(III) -C1-C4 -C1-C6 plant
(IV) -C1-C4 plant
Choose the correct answer from the options given below:
1.24 g of (molar mass 124 g mol ) is dissolved in 1 kg of water to form a solution with boiling point of 100.105 C, while 2.54 g of (molar mass 250 g mol ) in 2 kg of water constitutes a solution with a boiling point of 100.026 C. . Which of the following is correct?
(A) Weight of a substance is the amount of matter present in it.
(B) Mass is the force exerted by gravity on an object.
(C) Volume is the amount of space occupied by a substance.
(D) Temperatures below C are possible in Celsius scale, but in Kelvin scale negative temperature is not possible.
(E) Precision refers to the closeness of various measurements for the same quantity.
Choose the correct answer from the options given below:
In the given structure, number of and hybridized carbon atoms present respectively are: 
Match List - I with List - II.

Choose the correct answer from the options given below :
( A ) − ( I I ) , ( B ) − ( I ) , ( C ) − ( I I I ) , ( D ) − ( I V )
( A ) − ( I ) , ( B ) − ( I I ) , ( C ) − ( I V ) , ( D ) − ( I I I )
( A ) − ( I I ) , ( B ) − ( I ) , ( C ) − ( I V ) , ( D ) − ( I I I )
( A ) − ( I I ) , ( B ) − ( I I I ) , ( C ) − ( I ) , ( D ) − ( I V )

In the light of the above statements, choose the correct answer from the options given below:
The elemental composition of a compound is 54.2% C, 9.2% H, and 36.6% O. If the molar mass of the compound is 132 g/mol, the molecular formula of the compound is:
Match List - I with List - II.
For the reaction:

The correct order of set of reagents for the above conversion is :
One mole of an ideal gas expands isothermally and reversibly from to at , q and work done in the process respectively are : Given : and In
Let us consider a reversible reaction at temperature, T . In this reaction, both and were observed to have positive values. If the equilibrium temperature is , then the reaction becomes spontaneous at:
Given:
The for is _______ (nearest integer value).
In Carius method of estimation of halogen, 0.25 g of an organic compound gave 0.15 g of silver bromide (AgBr). The percentage of Bromine in the organic compound is ________________ × 10-1% (Nearest integer).
(Given: Molar mass of Ag is 108 and Br is 80 g mol-1)
Standard entropies of , and are 70, 50, and 110 J mol respectively. The temperature in Kelvin at which the reaction will be at equilibrium is (nearest integer):
A.
B.
C.
D.
E.
Choose the correct answer from the options given below:
In the given structure, number of and hybridized carbon atoms present respectively are:

For hydrogen atom, the orbital/s with lowest energy is/are:
Match List - I with List - II.

(A)-(IV), (B)-(III), (C)-(I), (D)-(II)
(A)-(III), (B)-(IV), (C)-(II), (D)-(I)
(A)-(I), (B)-(III), (C)-(II), (D)-(IV)
(A)-(III), (B)-(IV), (C)-(I), (D)-(II)
For the reaction:

The correct order of set of reagents for the above conversion is :
A perfect gas (0.1 mol) having R (independent of temperature) undergoes the above transformation from point 1 to point 4. If each step is reversible, the total work done (w) while going from point 1 to point 4 is ____ J (nearest integer) [Given : R = 0.082 L atm K ] 
A sample of n-octane (1.14 g) was completely burnt in excess of oxygen in a bomb calorimeter, whose heat capacity is 5 kJ K . As a result of combustion, the temperature of the calorimeter increased by 5 K. The magnitude of the heat of combustion at constant volume is ___
The molarity of a 70% (mass/mass) aqueous solution of a monobasic acid (X) is:
The possible number of stereoisomers for 5-phenylpent-4-en-2-ol is:
In Carius method of estimation of halogen, 0.25 g of an organic compound gave 0.15 g of silver bromide (AgBr). The percentage of Bromine in the organic compound is times 10^{-1}% (Nearest integer). (Given: Molar mass of Ag is 108 and Br is 80 g mol^{-1}
Given below are some nitrogen containing compounds:

Each of them is treated with HCl separately. 1.0 g of the most basic compound will consume ....... mg of HCl.
(Given Molar mass in g mol : C: 12, H: 1, O: 16, Cl: 35.5.)

Given data:
A. Expansion in single stage
B. Expansion in multi stages
C. Compression in single stage
D. Compression in multi stages
Choose the correct option.
at 600 K. The (in kJ ) for the reaction is :}

A. For water, magnitude of is more than the magnitude of .
B. The elevation in boiling point of water when a non-volatile solute is added to it is larger in magnitude than its depression in freezing point.
C. Osmotic pressure measurement is preferred over any other colligative property to determine molar mass of proteins and polymers.
D. The dimerised form of benzoic acid in benzene is C₆H₅-C-OH ⋯ O=C-C₆H₅

Statement-2: At constant volume as work done is zero, heat given to the chaotic motion is reflected by increase in temperature.
[Given: log 2 = 0.3, , , log 3 = 0.47]
Match the LIST-I with LIST-II. 
Choose the correct answer from the options given below:
5 C 100 C
Select the correct answer
Dissociation of a gas takes place according to the following chemical reaction. At equilibrium, the total pressure is at .
The standard Gibbs energy of formation of the involved substances is given below:
| Substance | (kJ mol ) |
|---|---|
The degree of dissociation of is given by
where ________ (Nearest integer).
[Given: , , . Assume degree of dissociation is not negligible.]
List-I:
(I) Isothermal reversible (1 mole ideal gas, T = 300K, 2dm to 20dm ) calculate
(II) Isothermal irreversible [3KPa, 1m to 3m ] calculate
(III) 1 mole gas undergoes constant pressure process in which change in temperature is 400K, C = 5R/2, will be
(IV) 1 mole ideal gas having C = 3R/2 and = 320K, calculate
List-II:
(A) 8.32
(B) 6
(C) 4
(D) 5.74
For Balanced chemical reaction
+ 6HCl 2AlCl + 3H (g)} \)
For the reaction
At 300 K and 1 atm pressure, the degree of dissociation of A gas at equilibrium is . Find .
If C(diamond) C(graphite) +
C(diamond) +
C(graphite) +
at constant temperature, then