JEE-MAIN SERIES Chemistry
States Of Matter
23 previous year questions.
Volume: 23 Ques
Yield: High
High-Yield Trend
2
2026 3
2023 6
2021 2
2019 2
2017 1
2016 2
2015 3
2014 2
2013 Chapter Questions 23 MCQs
01
PYQ 2013
easy
chemistry ID: jee-main
For gaseous state, if most probable speed is denoted by , average speed by and root square speed by , then for a large number of molecules, the ratios of these speeds are
1
2
3
4
02
PYQ 2013
hard
chemistry ID: jee-main
Which one of the following is the wrong assumption of kinetic theory of gases?
1
Momentum and energy always remain conserved.
2
Pressure is the result of elastic collision of molecules with the container's wall.
3
Molecules are separated by great distances compared to their sizes.
4
All the molecules move in straight line between collision and with same velocity.
03
PYQ 2014
medium
chemistry ID: jee-main
If Z is a compressibility factor, van der Waals equation at low pressure can be written as
1
2
3
4
04
PYQ 2014
easy
chemistry ID: jee-main
Van der Waal's equation for a gas is stated as, This equation reduces to the perfect gas equation, when ,
05
PYQ 2014
medium
chemistry ID: jee-main
The initial volume of a gas cylinder is 750.0 \,mL. If the pressure of gas inside the cylinder changes from Hg to 360.0 mm Hg, the final volume the gas will be :
1
1.750 L
2
3.60 L
3
4.032 L
4
7.50 L
06
PYQ 2015
medium
chemistry ID: jee-main
When does a gas deviate the most from its ideal behaviour ?
1
At low pressure and low temperature
2
At low pressure and high temperature
3
At high pressure and low temperature
4
At high pressure and high temperature
07
PYQ 2015
medium
chemistry ID: jee-main
The intermolecular interaction that is dependent on the inverse cube of distance between the molecules is
1
ion-ion interaction
2
ion-dipole interaction
3
London force
4
hydrogen bond
08
PYQ 2016
medium
chemistry ID: jee-main
At very high pressures, the compressibility factor of one mole of a gas is given by :
1
2
3
4
09
PYQ 2017
medium
chemistry ID: jee-main
At , the density of a certain gaseous molecule at bar is double to that of dinitrogen at bar. The molar mass of gaseous molecule is :
1
2
3
4
10
PYQ 2017
medium
chemistry ID: jee-main
Among the following, the incorrect statement is :
1
At low pressure, real gases show ideal behaviour
2
At very low temperature, real gases show ideal behaviour
3
At very large volume, real gases show ideal behaviour
4
At Boyle?s temperature, real gases show ideal behaviour
11
PYQ 2019
medium
chemistry ID: jee-main
An open vessel at is heated until two fifth of the air (assumed as an ideal gas) in it has escaped from the vessel. Assuming that the volume of the vessel remains constant, the temperature at which the vessel has been heated is :
1
2
3
4
12
PYQ 2019
medium
chemistry ID: jee-main
Points I, II and III in the following plot respectively correspond to ( most probable velocity)
1
2
3
4
13
PYQ 2021
medium
chemistry ID: jee-main
The interaction energy of London forces between two particles is proportional to r , where r is the distance between the particles. The value of x is :
1
3
2
-3
3
6
4
-6
14
PYQ 2021
medium
chemistry ID: jee-main
Which one of the following is the correct PV vs P plot at constant temperature for an ideal gas ? (P and V stand for pressure and volume of the gas respectively) 
1
A
2
B
3
C
4
D
15
PYQ 2021
medium
chemistry ID: jee-main
The unit of the van der Waals gas equation parameter 'a' in is:
1
kg m s
2
atm dm mol
3
dm mol
4
kg m s
16
PYQ 2021
medium
chemistry ID: jee-main
Two flasks I and II shown below are connected by a valve of negligible volume. When the valve is opened, the final pressure of the system in bar is . The value of x is _________. (Integer answer)
[Assume - Ideal gas; 1 bar = 10 Pa; Molar mass of N = 28.0 g mol ; R=8.31 J mol K ] 
17
PYQ 2021
medium
chemistry ID: jee-main
The volume occupied by 4.75 g of acetylene gas at 50 °C and 740 mmHg pressure is _______ L. (Rounded off to the nearest integer) [Given ]
18
PYQ 2021
medium
chemistry ID: jee-main
A car tyre is filled with nitrogen gas at 35 psi at 27 C. It will burst if pressure exceeds 40 psi. The temperature in C at which the car tyre will burst is ___________
19
PYQ 2023
hard
chemistry ID: jee-main
Number of molecules and moles in 2.8375 litre of O2 at STP.
20
PYQ 2023
medium
chemistry ID: jee-main
Based on the given figure, the number of correct statement/s is/are _______
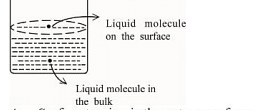
A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system

A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system
21
PYQ 2023
hard
chemistry ID: jee-main
A metal chloride contains 55% by mass of chlorine. 100 mL of vapours gives 0.57 gm of chlorine at STP. Calculate the molecular mass of metal chloride. (Nearest integer)
22
PYQ 2026
medium
chemistry ID: jee-main
Given below are two statements: Statement I: Sublimation is a purification technique that is used to separate those solid substances which change from solid to vapour state without passing through liquid state. Statement II: If external atmospheric pressure is reduced, then boiling point of a substance is decreased. In the light of the above statements, choose the correct option.
1
Both statement I and statement II are correct
2
Both statement I and statement II are incorrect
3
Statement I is correct but statement II is incorrect
4
Statement I is incorrect but statement II is correct
23
PYQ 2026
medium
chemistry ID: jee-main
Statement-1: Sublimation is a purification technique that is used to separate those solid substances which change from solid to vapor state without passing through liquid state.
Statement-2: If external atmospheric pressure is reduced, then boiling point of substance decreases.
Which of the following is correct?
Statement-2: If external atmospheric pressure is reduced, then boiling point of substance decreases.
Which of the following is correct?
1
Statement-1 and Statement-2 both are correct.
2
Statement-1 and Statement-2 both are incorrect.
3
Statement-1 is correct but Statement-2 is incorrect.
4
Statement-1 is incorrect but Statement-2 is correct.
