Chemical Reactions
83 previous year questions.
High-Yield Trend
Chapter Questions 83 MCQs
The order of stability of the following carbocations

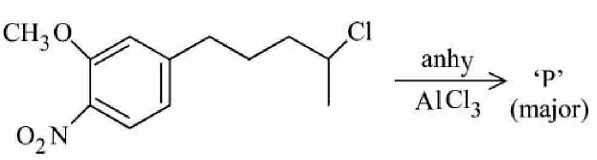
The major product of the following reaction is









The amount of HNO3 required to produce 110.0 g of KNO3 is
(Given : Atomic masses of H, O, N and K are 1, 16, 14 and 39 respectively.)
Consider the above reaction and predict the major product.










quinonoid form
heterocyclic form
phenolic form
benzenoid form
For the decomposition of azomethane, CH3N2CH3(g) → CH3CH3(g) + N2(g), a first-order reaction, the variation in partial pressure with time at 600 K is given as
The half-life of the reaction is ______ × 10–5s. [Nearest integer]
-
Choose the correct answer from the options given below :
Arrange the following in the decreasing order of their covalent character :
(A) LiCl
(B) NaCl
(C) KCl
(D) CsCl
Choose the most appropriate answer from the options given below :
Statement I: Classical smog occurs in cool humid climate. It is a reducing mixture of smoke, fog and sulphur dioxide.
Statement II: Photochemical smog has components, ozone, nitric oxide, acrolein, formaldehyde, PAN etc.
In the light of the above statements, choose the most appropriate answer from the options given below.

In the above reaction product B is:
Product B is








‘




A.
B.
C.
D.









Identify and in the following reaction (Equation not balanced)
Consider the following reaction sequence:




When 2 gm magnesium reasts with excess of HCl and H2 gas is produced then the volume of H2 gas produced is ____ × 10–2 liter at STP. (Nearest Integer)









Na2O + H2O → 2X
Cl2O7 + H2O → 2Y
For the given reaction, if the initial pressure is 450 mm Hg and the pressure at time t is 720 mm Hg at a constant temperature T and constant volume V. The fraction of A(g) decomposed under these conditions is . The value of x is _____ . (nearest integer)

The Product ‘X’ is:




Formation of photochemical smog involves the following reaction in which A, B, and C are respectively:
Choose the correct answer from the options given below:
NO, O, and O3
N2O and NO
N, O2, and O3
O, NO, and NO3










In the above conversion the correct sequence of reagents to be added is






The product 'B' is




A. They bind to the receptor site.
B. Get transferred inside the cell for their action.
C. Inhibit the natural communication of the body.
D. Mimic the natural messenger.
Choose the correct answer from the options given below:





Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: H2Te is more acidic than H2S.
Reason R: Bond dissociation enthalpy of H2Te is lower than H2S.
In light of the above statements, choose the most appropriate from the options given below:






A and B respectively are:













Choose the correct answer from the options given below.






(Given atomic number of Mn is 25)

Consider the above chemical reaction. Product "A" is:




| List-I Reaction | List-II Type of redox reaction |
|---|---|
| (A) N2(g) + O2(g) → 2NO(g) | (I) Decomposition |
| (B) 2Pb (NO3)2(s) → 2PbO(s) + 4NO2(g) + O2(g) | (II) Displacement |
| (C) 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) | (III) Disproportionation |
| (D) 2NO2(g) + 2OH-(aq) → NO2-(aq) + NO3-(aq) + H2O(l) | (IV) Combination |
In Carius method for estimation of halogens, 180 mg of an organic compound produced 143.5 mg of AgCl. The percentage composition of chlorine in the compound is ___________%. [Given: Molar mass in g mol of Ag = 108, Cl = 35.5]
Statement (II): The boiling points of alcohols and phenols are higher in comparison to other classes of compounds such as ethers and haloalkanes. In the light of the above statements, choose the correct answer from the options given below:
Consider the following reaction occurring in the blast furnace. ‘x’ kg of iron is produced when kg and kg CO are brought together in the furnace.
The value of ‘x’ is __________ (nearest integer).
X g of benzoic acid on reaction with aqueous release that occupied 11.2 L volume at STP. X is ________ g.
Match the list-I with list-II 











