JEE-MAIN SERIES Chemistry
Ideal Gas Equation
14 previous year questions.
Volume: 14 Ques
Yield: Medium
High-Yield Trend
5
2023 6
2022 1
2019 1
2016 1
2014 Chapter Questions 14 MCQs
01
PYQ 2014
medium
chemistry ID: jee-main
The ratio mass of oxygen and nitrogen of a particular gaseous
mixture is The ratio of number of their molecule is
1
2
3
4
02
PYQ 2016
medium
chemistry ID: jee-main
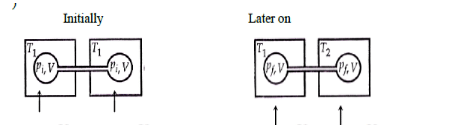
Two closed bulbs of equal volume containing an ideal gas initially at pressure and temperature are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to . The final pressure is :
1
2
3
4
03
PYQ 2019
medium
chemistry ID: jee-main
One mole of an ideal gas passes through a process where pressure and volume obey the relation .Here and are constants. Calculate the change in the temperature of the gas if its volume changes from to .
1
2
3
4
04
PYQ 2022
medium
chemistry ID: jee-main
For a real gas at 25°C temperature and high pressure (99 bar) the value of the compressibility factor is 2, so the value of van der Waal’s constant ‘b’ should be ______ \times 10–2 L mol–1 (Nearest integer).
(Given R=0.083 L bar K-1 mol-1)
(Given R=0.083 L bar K-1 mol-1)
05
PYQ 2022
easy
chemistry ID: jee-main
A gas (Molar mass = 280 g mol–1) was burnt in excess O2 in a constant volume calorimeter and during combustion the temperature of calorimeter increased from 298.0 K to 298.45 K. If the heat capacity of calorimeter is 2.5 kJ K–1 and enthalpy of combustion of gas is 9 kJ mol–1 then amount of gas burnt is __________g. (Nearest Integer).
06
PYQ 2022
medium
chemistry ID: jee-main
2.0 g of H2 gas is adsorbed on 2.5 g of platinum powder at 300 K and 1 bar pressure. The volume of the gas adsorbed per gram of the adsorbent is _____ mL.
(Given : R= 0.083 L bar K–1 mol–1)
(Given : R= 0.083 L bar K–1 mol–1)
07
PYQ 2022
medium
chemistry ID: jee-main
100 g of an ideal gas is kept in a cylinder of 416 L volume at 27°C under 1.5 bar pressure. The molar mass of the gas is ________ g mol–1. (Nearest integer). (Given : R = 0.083 L bar K–1 mol–1)
08
PYQ 2022
medium
chemistry ID: jee-main
An evacuated glass vessel weighs 40.0 g when empty, 135.0 g when filled with a liquid of density 0.95 g mL–1 and 40.5 g when filled with an ideal gas at 0.82 atm at 250 K. The molar mass of the gas in g mol–1 is: (Given : R = 0.082 L atm K–1 mol–1)
1
35
2
50
3
75
4
125
09
PYQ 2022
medium
chemistry ID: jee-main
Given below are the critical temperatures of some of the gases :
| Gas | Critical temperature (K) |
|---|---|
| He | 5.2 |
| CH4 | 190.0 |
| CO2 | 304.2 |
| NH3 | 405.5 |
The gas showing least adsorption on a definite amount of charcoal is
1
He
2
CH4
3
CO2
4
NH3
10
PYQ 2023
hard
chemistry ID: jee-main
A sealed flask with a capacity of contains of propane gas The flask is so weak that it will burst if the pressure becomes The minimum temperature at which the flask will burst is ______ [Nearest integer]
(Given: Atomic masses of and are and respectively) (Assume that propane behaves as an ideal gas)
11
PYQ 2023
hard
chemistry ID: jee-main
The total pressure of a mixture of non-reacting gases and in a vessel is of The partial pressure of the gas is ____ of (Nearest Integer)(Given : molar mass and )
12
PYQ 2023
easy
chemistry ID: jee-main
When a 60 W electric heater is immersed in a gas for 100s in a constant volume container with adiabatic walls, the temperature of the gas rises by 5°C. The heat capacity of the given gas is_____J K-1 (Nearest integer).
13
PYQ 2023
hard
chemistry ID: jee-main
One mole of an ideal gas at 350K is in a 2.0 L vessel of thermally conducting walls, which are in contact with the surroundings. It undergoes isothermal reversible expansion from 2.0L to 3.0L against a constant pressure of 4 atm. The change in entropy of the surroundings (ΔS) is JK (Nearest integer) Given: R = 8.314 J K-1 Mol-1.
14
PYQ 2023
medium
chemistry ID: jee-main
A certain quantity of real gas occupies a volume of 30.15 dm3 at 100 atm and 500 K when its compressibility factor is 1.07. Its volume at 300 atm and 300K (When its compressibility factor is 1.4) is _____ x 10-4 dm3 (Nearest integer)