Coordination Compounds
170 previous year questions.
High-Yield Trend
Chapter Questions 170 MCQs




Arrange the following Cobalt complexes in the order of increasing Crystal Field Stabilization Energy (CFSE) value. 
[ Assume no volume change on adding ]
Statement I : [Mn(CN) ] , [Fe(CN) ] and [Co(C O ) ] are d sp hybridised.
Statement II : [MnCl ] and [FeF ] are paramagnetic and have 4 and 5 unpaired electrons, respectively.
Choose the correct option.
(i) [FeF ] (ii) [Co(NH ) ] (iii) [NiCl ] (iv) [Cu(NH ) ]
Statement I: The identification of Ni is carried out by dimethyl glyoxime in the presence of NH OH.
Statement II: The dimethyl glyoxime is a bidentate neutral ligand.
| List-I (Complex) | List-II (Hybridization) | ||
|---|---|---|---|
| A. | Ni(CO)4 | I. | sp3 |
| B. | [Ni(CN)4]2- | II. | sp3d2 |
| C. | [Co(CN)6]3- | III. | d2sp3 |
| D. | [CoF6]3- | IV. | dsp2 |
(en – ethylenediamine;H2N−CH2−CH2−NH2)
[Cu(H2O)6]SO4
[Cu(en)(H2O)4]SO4
cis−[Cu(en)2Cl2]
trans−[Cu(en)2Cl2]
Consider the following metal complexes:
[Co(NH3)6]3+
[CoCl(NH3)5]2+
[Co(CN)6]3–
[Co(NH3)5(H2O)]3+
The spin-only magnetic moment value of the complex that absorbs light with shortest wavelength is _______ B.M. (Nearest integer)
| List-I (Compound) | List-II (Shape) |
|---|---|
| (A) | (I) bent |
| (B) | (II) square pyramidal |
| (C) | (III) trigonal bipyramidal |
| (D) | (IV) octahedral |
Statement I : In molecule the covalent radius is double of the atomic radius of chlorine.
Statement II : Radius of anionic species is always greater than their parent atomic radius.
Choose the most appropriate answer from options given below:
| List-I (Processes/ Reactions) | List-II (Catalyst) |
|---|---|
| (A) | (I) |
| (B) | (II) |
| (C) | (III) |
| (D) Vegetable oil | (IV) Vegetable ghee(s) |
(1)
(2)
(3) (high spin)
(4)
[Fe(CN)6]3–
[Ti(CN)6]3–
[Ni(CN)4]2–
[Co(CN)6]3–
Among the given complexes, number of paramagnetic complexes is_______.
Given below are two statements.
Statement-I: In CuSO4.5H2O, Cu-O bonds are present.
Statement-II: In CuSO4.5H2O, ligands coordinating with Cu(II) ion are O-and S-based ligands.
In the light of the above statements, choose the correct answer from the options given below:
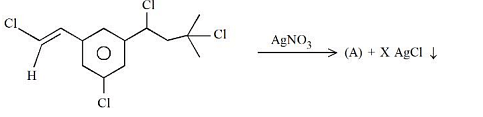
For the reaction given below:
If two equivalents of AgCl precipitate out, then the value of x will be____.
| List-I Coordination entity | List-II Wavelength of light absorbed in nm | ||
| A. | I. | 310 | |
| B. | II. | 475 | |
| C. | III. | 535 | |
| D. | IV. | 600 | |
Spin only magnetic moment of Fe in [Fe(H2O)6] 3+ and [Fe(CN)6] 3– complexes respectively is:
, Diamagnetic
, Paramagnetic
, Diamagnetic
, Paramagnetic
Maximum number of isomeric monochloro derivatives which can be obtained from 2,2,5,5-tetramethylhexane by chlorination is ________
Find the spin magnetic moment ratio for complexes &
[Pt(NH3)3CI3]+
[Pt(en)3]4+
[Pt(en)2Cl2]2+
[Pt(en)2(NH3)2]4+
The hybridization and magnetic behaviour of cobalt ion in complex, respectively is
and paramagnetic
and paramagnetic
and diamagnetic
and diamagnetic
| List I (Complex) | List II (Hybridization) | ||
| A. | Ni(CO)4 | I. | sp3 |
| B. | [Cu(NH3)4]2+ | II. | dsp2 |
| C. | [Fe(NH3)6]2+ | III. | sp3d2 |
| D. | [Fe(H2O)6]2+ | IV. | d2sp3 |
A ⇒ Spin only magnetic moment of [Fe(CN)6]–3 is 1.73 B.M. and [Fe(H2O)6]+3 is 5.92 B.M.
R ⇒ In both cases Fe have +3 oxidation state
In the light of the above statements, write the detailed answer:
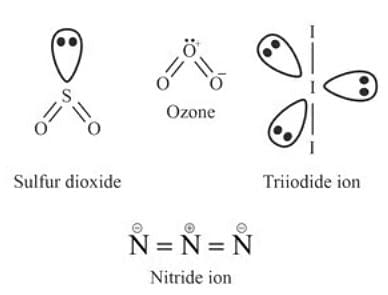
SO2, O3, I3-, N3-
Assertion: Magnetic moment of [Fe(H2O)6]3+ is 5.92 BM and that of [Fe(CN)6]3– is 1.73 BM
Reason: Oxidation state of Fe in both the complexes is +3.
In the light of the above statements, choose the correct answer from the options given below
Both Assertion and Reason are correct and Reason is the correct explanation of Assertion
Both Assertion and Reason are correct but Reason is not the correct explanation of Assertion
Reason is correct but Assertion is not correct
Reason is incorrect but Reason is correct

Calculate mass of Tollen’s Reagent Required?
18.70 kg
37.40 kg
9.35 kg
55.10 kg
Assertion A: [CoCl(NH3)]2– absorbs at lower wavelength of light with respect to [Co(NH3)5(H2O) 3–
Reason R: It is because the wavelength of the light absorbed depends on the oxidation state of the metal ion.
In the light of the above statements, choose the correct answer from the options given below:
Both A and R are true and R is the correct explanation of A
A is true but R is false
(Given At.no. Fe: 26, Co:27)
lead sulphate
lead nitrate
lead chromate
lead iodide
Number of ambidentate ligands in given complex [M(en)(SCN)4] :
Spin only magnetic moment of is B.M. and is B.M.
In both cases Fe have oxidation state.
In the light of the above statements, write the detailed answer:
(A)
(B)
(C)
(D)
The correct order of A, B, C, and D in terms of wavenumber of light absorbed is:

Choose the correct answer from the options given below:
A. The strength of anionic ligands can be explained by crystal field theory.
B. Valence bond theory does not give a quantitative interpretation of kinetic stability of coordination compounds.
C. The hybridization involved in formation of complex is .
D. The number of possible isomer(s) of is one.
Choose the correct answer from the options given below:
A. The chromate ion is square planar.
B. Dichromates are generally prepared from chromates.
C. The green manganate ion is diamagnetic.
D. Dark green coloured disproportionates in a neutral or acidic medium to give permanganate.
E. With increasing oxidation number of transition metal, ionic character of the oxides decreases.
Choose the correct answer from the options given below:
| LIST-I | LIST-II |
|---|---|
| A. K2[Ni(CN)4] | I. sp3 |
| B. [Ni(CO)4] | II. sp3d2 |
| C. [Co(NH3)6]Cl3 | III. dsp2 |
| D. Na3[CoF6] | IV. d2sp3 |
Choose the correct answer from the options given below:
A Ethane-1,2-diamine is a chelating ligand.
B Metallic aluminium is produced by electrolysis of aluminium oxide in presence of cryolite.
C Cyanide ion is used as a ligand for leaching of silver.
D Phosphine acts as a ligand in Wilkinson catalyst.
E The stability constants of and are similar to EDTA complexes.
P =
Q =
R =
The correct order of the complex ions, according to their spin-only magnetic moment values (in B.M.) is:
The structure of the major product formed in the following reaction is:





The steam volatile compounds among the following are:

The structure of the major product formed in the following reaction is: 




Statement I: 3-phenylpropene reacts with and gives secondary alkyl bromide having a chiral carbon atom as the major product.
Statement II: Aryl chlorides and aryl cyanides can be prepared by Sandmeyer reaction as well as Gattermann reaction.
In the light of the above statements, choose the correct answer from the options given below
Statement I: Methane can be prepared by decarboxylation of sodium ethanoate, Kolbe's electrolysis of sodium acetate and reaction of with water.
Statement II: Methane cannot be prepared from unsaturated hydrocarbons and by Wurtz reaction.
In the light of the above statements, choose the correct answer from the options given below
B. The complex is white in colour.
C. The calculated spin-only magnetic moment of the complex is BM.
D. The calculated CFSE (Crystal Field Stabilization Energy) of Ni in this complex is .
E. The geometrical arrangement of ligands in this complex is similar to that in Ni(CO) .
Choose the correct answer from the options given below:
Statement I: The number of paramagnetic species among , , and is 3.
Statement II: is the correct order in terms of number of unpaired electrons.
Choose the correct answer from the options given below:
Both Statement I and Statement II are true
| Column-I (Complex compound) | Column-II ( (CFSE) ) |
| (i) | (P) 17000 |
| (ii) | (Q) 15000 |
| (iii) | (R) 12000 |
| (iv) | (S) 20000 |
mol of is formed
(B)
(C)
(D)
Select correct order of spin-only magnetic moment among above complexes.
Given below are two statements:
Statement I: The number of species among , , , , , and , that have tetrahedral geometry is 3.
Statement II: In the set , , , , all the molecules have incomplete octet around the central atom.
In the light of the above statements, choose the correct answer from the options given below:
Consider a weak base of . mL of M HCl and mL of M weak base are mixed to make mL of a buffer of pH at . The values of and respectively are
Given below are two statements regarding conformations of n-butane. Choose the correct option. 
Statement I: Hybridisation, shape and spin only magnetic moment of is , octahedral and BM respectively.
Statement II: Geometry, hybridisation and spin only magnetic moment values (BM) of the ions , and respectively are square planar, tetrahedral, octahedral; , , and , , .
In the light of the above statements, choose the correct answer from the options given below:
Both Statement I and Statement II are false
Identify the CORRECT set of details from the following:
A. : Inner orbital complex; hybridized
B. : Outer orbital complex; hybridized
C. : Outer orbital complex; hybridized
D. : Outer orbital complex; hybridized
E. : Inner orbital complex; hybridized}
Statement I : Crystal Field Stabilization Energy (CFSE) of is greater than that of .
Statement II : Potassium ferricyanide has a greater spin-only magnetic moment than sodium ferrocyanide.
In the light of the above statements, choose the correct answer from the options given below :}
(Given: Molar mass in g mol Cr: 52; Cl: 35.5; Ag:108; O:16; H:1)
Statement I: Among [Cu(NH₃)₄]²⁺, [Ni(en)₃]²⁺, [Ni(NH₃)₆]²⁺ and [Mn(H₂O)₆]²⁺, [Mn(H₂O)₆]²⁺ has the maximum number of unpaired electrons.
Statement II: The number of pairs among {[NiCl₄]²⁻, [Ni(CO)₄], {[NiCl₄]²⁻, [Ni(CN)₄]²⁻} and {[Ni(CO)₄], [Ni(CN)₄]²⁻} that contain only diamagnetic species is two.
In the light of the above statements, choose the correct answer from the options given below:}
Consider the following conformations: 
Statement-I: I is more stable than I .
Statement-II: As dihedral angle increases, stability decreases.
Statement-I: Crystal field stabilisation energy (magnitude) of is greater than .
Statement-II: Order of bond energy is .
In the light of the above statements, choose the correct option.
Consider the following complexes:
(A) [Co(CN) ]
(B) [Co(NH ) H O]
(C) [Co(H O) ]
(D) [CoF ]
The wavelength absorbed by the above complexes are in the order:

Arrange the given metal ions in increasing order of number of unpaired electrons in the low spin complexes formed by
Statement-II: [CoCl ] has higher crystal field splitting energy than [CoBr ]
(B) It is readily soluble in water at pH = 9.
(C) Central metal ion has two unpaired electrons.
(D) It has four, 5-membered metal containing rings.