Organic Chemistry
458 previous year questions.
High-Yield Trend
Chapter Questions 458 MCQs












Two compounds I and II are eluted by column chromatography (adsorption of I > II). Which one of following is a correct statement ?





In the following sequence of reactions, the final product D is : 
Which one of the following compounds will give orange precipitate when treated with 2,4-dinitrophenyl hydrazine ? 

The correct structures of trans-[NiBr₂(PPh₃)₂] and meridional-[Co(NH₃)₃(NO₂)₃], respectively, are : 
What is the correct sequence of reagents used for converting nitrobenzene into m-dibromobenzene ? 
What is the final product (major) 'A' in the given reaction ? 

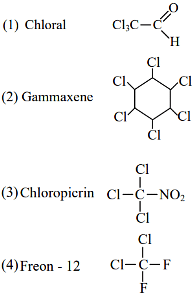
(a) Alcoholic potassium hydroxide
(b) Pd/BaSO₄
(c) BHC (Benzene hexachloride)
(d) Polyacetylene List - II (Use/Preparation/Constituent)
(i) electrodes in batteries
(ii) obtained by addition reaction
(iii) used for β-elimination reaction
(iv) Lindlar's Catalyst
Statement I : Aniline is less basic than acetamide.
Statement II : In aniline, the lone pair of electrons on nitrogen atom is delocalised over benzene ring due to resonance and hence less available to a proton.
Choose the most appropriate option :
The correct sequence of correct reagents for the following transformation is : 
(Atomic Mass of Ba=137 u, S=32 u, O=16 u)
The major product of the following reaction is: 
Statement I : Ethyl pent-4-yn-oate on reaction with CH MgBr gives a 3 -alcohol.
Statement II : In this reaction one mole of ethyl pent-4-yn-oate utilizes two moles of CH MgBr.
In the light of the above statements, choose the most appropriate answer from the options given below :
.
The value of is ________. (Integer answer)
Assertion (A) : A simple distillation can be used to separate a mixture of propanol and propanone.
Reason (R) : Two liquids with a difference of more than in their boiling points can be separated by simple distillations.
In the light of the above statements, choose the most appropriate answer from the options given below :
Choose the correct name for compound given below : 
In the following molecule,
Hybridisation of Carbon a, b and c respectively are :
Statement I : Hyperconjugation is a permanent effect.
Statement II : Hyperconjugation in ethyl cation involves the overlapping of bond with empty orbital of the other carbon.
Choose the correct option :
Consider the above reaction, the major product "P" formed is : 
Which one among the following resonating structures is not correct ? 

(a) Chlorophyll
(b) Vitamin - B₁₂
(c) Anticancer drug
(d) Grubbs catalyst List - II
(i) Ruthenium
(ii) Platinum
(iii) Cobalt
(iv) Magnesium
Which of the following compound gives pink colour on reaction with phthalic anhydride in conc. followed by treatment with ? 
Which one of the following compounds is non-aromatic ? 

The correct order of acid character of the following compounds is :
I: Phenol, II: p-Nitrobenzoic acid, III: Benzoic acid, IV: p-Toluic acid 
(A) Sulphanilic acid (B) Picric acid (C) Aspirin (D) Ascorbic acid
The correct order of stability of given carbocations is : 
Consider the above reaction and identify the Product P : 

(Atomic masses of Ag and Cl are 107.87 and 35.5 respectively)




| List-I(Mixture) | List-II(Purification Process) |
| (A) Chloroform & Aniline | (I) Steam distillation |
| (B) Benzoic acid & Napthalene | (II) Sublimation |
| (C) Water & Aniline | (III) Distillation |
| (D) Napthalene & Sodium chloride | (IV) Crystallisation |
The number of sp3 hybridised carbons in an acyclic neutral compound with molecular formula C4H5N is ________.
Ofloxacin, Penicillin G, Terpineol, Salvarsan.





Benzyl isocyanide can be obtained by:
Choose the correct answer from the options given below:
A and D
Only B
B and C
A and B
In Dumas method for the estimation of , the sample is heated with copper oxide and the gas evolved is passed over :
In the following halogenated organic compounds the one with maximum number of chlorine atoms in its structure is :
On complete combustion, of an organic compound gave of The of carbon in the organic compound is(Nearest integer)
PbSO4
Pb(NO3)2
PbCrO4
PbI2
Which of the following compounds would give the following set of qualitative analysis?
- Fehling's Test: Positive
- Na fusion extract upon treatment with sodium nitroprusside gives a blood-red colour but not prussian blue








What is the chemical formula of freon gas?
C2Cl2F4
C2F2H4
CHF3
CCl2F2
Match List I with List II:
A – II, B – I, C – IV, D – III
A – I, B – III, C – II, D – I
A – I, B – II, C – IV, D – III
A – IV, B – I, C – III, D – II

















Assertion (A): CH2 = CH – CH2 – Cl is an example of allyl halide
Reason (R): Allyl halides are the compounds in which the halogen atom is attached to sp2 hybridised carbon atom.
In the light of the two above statements, choose the most appropriate answer from the options given below




Statement I: Bromination of phenol in a solvent with low polarity such as CHCl or CS requires a Lewis acid catalyst.
Statement II: The Lewis acid catalyst polarizes the bromine to generate .
In light of the above statements, choose the correct answer from the options given below:
Assertion A: The stability order of oxidation state of Ga, In, and Tl is .
Reason R: The inert pair effect stabilizes the lower oxidation state down the group.
In the light of the above statements, choose the correct answer from the options given below:





- Column chromatography
- Thin layer chromatography
- Paper chromatography





Statement I : Picric acid is 2, 4, 6-trinitrotoluene.
Statement II : Phenol-2, 4-disulphuric acid is treated with conc. HNO3 to get picric acid.
In the light of the above statement, choose the most appropriate answer from the options given below:
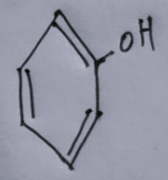
Assertion (A) : Haloalkanes react with KCN to form alkyl cyanides as a main product while with AgCN form isocyanide as the main product.
Reason (R) : KCN and AgCN both are highly ionic compounds. In the light of the above statement
Choose the most appropriate answer from the options given below:

Alkaline oxidative fusion of gives “A” which on electrolytic oxidation in alkaline solution produces B. A and B respectively are:

Product A and B formed in the following set of reactions are:









A. Glycerol is purified by vacuum distillation because it decomposes at its normal boiling point
B. Aniline can be purified by steam distillation as aniline is miscible in water.
C. Ethanol can be separated from ethanol water mixture by azeotropic distillation because it forms azeotrope.
D. An organic compound is pure, if mixed M.P. is remained same.
Choose the most appropriate answer from the options given below :






(consider complete conversion)

Statement II: is colourless.
Compounds: Benzaldehyde, Acetaldehyde, Acetone, Acetophenone, Methanal, 4-nitrobenzaldehyde, cyclohexane carbaldehyde.












Choose the most appropriate answer from the options given below:
| List-I (Compound) | List-II (Uses) |
|---|---|
| (A) Iodoform | (I) Fire extinguisher |
| (B) Carbon tetrachloride | (II) Insecticide |
| (C) CFC | (III) Antiseptic |
| (D) DDT | (IV) Refrigerants |
toluene, nitrobenzene, xylene, cumene, aniline, chlorobenzene, m-nitroaniline, m-dinitrobenzene

In the above reaction product ’P’ is




One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: value of phenol is 10.0 while that of ethanol is 15.9.
Reason R: Ethanol is stronger acid than phenol.
In the light of the above statements, choose the correct answer from the options given below:
One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: Alcohols react both as nucleophiles and electrophiles.
Reason R: Alcohols react with active metals such as sodium, potassium and aluminum to yield corresponding alkoxides and liberate hydrogen.
In the light of the above statements, choose the correct answer from the options given below:




Statement I: Nitration of benzene involves the following step –

Statement II: Use of Lewis base promotes the electrophilic substitution of benzene. In the light of the above statements.
Choose the most appropriate answer from the options given below :
(A)
(B)
(C)
The increasing order of boiling point is: Choose the correct answer from the options given below:
Statement I: D-glucose pentaacetate reacts with 2,4-dinitrophenylhydrazine.
Statement II: Starch, on heating with concentrated sulfuric acid at 100°C and 2-3 atmosphere pressure, produces glucose.
In the light of the above statements, choose the correct answer from the options given below:




Which among the following react with Hinsberg's reagent?

Choose the correct answer from the options given below:
Given below are two statements:

involving lone pair of electrons on nitrogen.
The compounds which give positive Fehling's test are:
Choose the CORRECT answer from the options given below:
Consider the following amino acid: 
Which of the following options contain the correct structure of (A) and (B)?




Match List-I with List-II
Choose the correct answer from the options given below :
(iso-Bu = isobutyl, sec-Bu = sec-butyl, nPr = n-propyl, tBu = tert-butyl, Et = ethyl)




The correct stability order of the following species/molecules is:

The most stable carbocation from the following is:




Which among the following compounds give yellow solid when reacted with NaOI/NaOH? 
Choose the correct answer from the options given below:
(B)
(C)
(D)
List - II: (I) & diamagnetic
(II) & paramagnetic
(III) & diamagnetic
(IV) & paramagnetic
Choose the correct answer from the options given below:
Identify correct statement/s:
(A) and are activating groups.
(B) and are meta directing groups.
(C) and are meta directing groups.
(D) Activating groups act as ortho- and para-directing groups.
(E) Halides are activating groups.
Choose the correct answer from the options given below:
Choose the correct set of reagents for the following conversion:
The IUPAC name of the following compound is:
Match the Compounds (List - I) with the appropriate Catalyst/Reagents (List - II) for their reduction into corresponding amines. 
( A ) − ( I I I ) , ( B ) − ( I V ) , ( C ) − ( I I ) , ( D ) − ( I )
( A ) − ( I I ) , ( B ) − ( I V ) , ( C ) − ( I I I ) , ( D ) − ( I )
( A ) − ( I I ) , ( B ) − ( I ) , ( C ) − ( I I I ) , ( D ) − ( I V )
( A ) − ( I I I ) , ( B ) − ( I I ) , ( C ) − ( I V ) , ( D ) − ( I )
The major product of the following reaction is:





Consider the following sequence of reactions : 
Molar mass of the product formed (A) is ______ g mol .
0.01 mole of an organic compound (X) containing 10% hydrogen, on complete combustion, produced 0.9 g H₂O. Molar mass of (X) is ___________g mol .
The observed and normal molar masses of compound MX2 are 65.6 and 164 respectively. The percent degree of ionisation of MX2 is ________________% (Nearest integer).
The sequence from the following that would result in giving predominantly 3, 4, 5 -Tribromoaniline is :









Match List - I with List - II:
List - I | List - II |
(A) Adenine | (I) |
(B) Cytosine | (II) |
(C) Thymine | (III) |
(D) Uracil | (IV) |
Choose the correct answer from the options given below:
A. Iodoform Reaction
B. Cannizzaro Reaction
C. Aldol Condensation
D. Tollen's Test
E. Clemmensen Reduction
Choose the correct answer from the options given below:
The total number of structural isomers possible for the substituted benzene derivatives with the molecular formula is __

Choose the correct option for structures of A and B, respectively:









The correct order of stability of following carbocations is: 
The products formed in the following reaction sequence are: 




In the following substitution reaction:




Statement I: Ozonolysis followed by treatment with Zn, H O of cis-2-butene gives ethanal.
Statement II: The product obtained by ozonolysis followed by treatment with Zn, H O of 3, 6-dimethyloct-4-ene has no chiral carbon atom.
In the light of the above statements, choose the correct answer from the options given below.
When  undergoes intramolecular aldol condensation, the major product formed is:
undergoes intramolecular aldol condensation, the major product formed is:





A shows positive Lassaigne's test for N and its molar mass is 121.
B gives effervescence with aqueous NaHCO3.
C gives fruity smell.
Identify A, B, and C from the following.




What is the correct IUPAC name of the following compound? 
Match the LIST-I with LIST-II

Choose the correct answer from the options given below:
What is the correct IUPAC name of

Given below are two statements :
Statement I : Hyperconjugation is not a permanent effect.
Statement II : In general, greater the number of alkyl groups attached to a positively charged C-atom, greater is the hyperconjugation interaction and stabilization of the cation.
In the light of the above statements, choose the correct answer from the options given below
In the following series of reactions identify the major products A and B respectively.




The major product (P) in the following reaction is :





In which pairs, the first ion is more stable than the second?

Identify the structure of the final product (D) in the following sequence of the reactions :
Total number of hybridised carbon atoms in product D is _____.




Match List-I with List-II and select the correct option: 
The correct IUPAC name of the product is: 
The correct sequence of reagents to be added for the following conversion is: 








The major product (A) formed in the following reaction sequence is









The least acidic compound, among the following is 




Identify [A], [B], and [C], respectively in the following reaction sequence : 




Match List - I with List - II.

Statement I: Experimentally determined oxygen-oxygen bond lengths in the are found to be the same and the bond length is greater than that of a (double bond) but less than that of a single bond.
Statement II: The strong lone pair-lone pair repulsion between oxygen atoms is solely responsible for the fact that the bond length in ozone is smaller than that of a double bond but more than that of a single bond .
In light of the above statements, choose the correct answer from the options given below:
For the thermal decomposition of at constant volume, the following table can be formed, for the reaction mentioned below: Given: Rate constant for the reaction is .

Identify the correct statements from the following 
Choose the correct answer from the options given below
Predict the major product of the following reaction sequence: 




An organic compound (X) with molecular formula is not readily oxidised. On reduction it gives which reacts with HBr to give a bromide (Z) which is converted to Grignard reagent. This Grignard reagent on reaction with (X) followed by hydrolysis give 2,3-dimethylbutan-2-ol. Compounds (X), (Y) and (Z) respectively are:
Statement II: {CH_3-C(-CH_3)(-CH_3)-CH_2-Cl}
will not undergo reaction very easily though it is a primary halide. In the light of the above statements, choose the most appropriate answer from the options given below:
Given below are two statements :
In the light of the above statements, choose the most appropriate answer from the options given below :
The number of optically active products obtained from the complete ozonolysis of the given compound is :




The product (P) formed in the following reaction is: 




i)
ii)
iii)
11.25 mg of chlorobenzene will produce mg of product B. Given molar mass of C, H, O, N, and Cl as 12, 1, 16, 14, and 35.5 g mol , respectively, the value of is:

A molecule (P) on treatment with acid undergoes rearrangement and gives (Q). (Q) on ozonolysis followed by reflux under alkaline condition gives (R). The structure of (R) is given below. The structure of (P) is:






2,2,3,3-Tetramethylpentane
Given below are two statements:
Statement I: Experimentally determined oxygen-oxygen bond lengths in the are found to be the same and the bond length is greater than that of a (double bond) but less than that of a single bond.
Statement II: The strong lone pair-lone pair repulsion between oxygen atoms is solely responsible for the fact that the bond length in ozone is smaller than that of a double bond but more than that of a single bond .
In light of the above statements, choose the correct answer from the options given below:
Given below are two statements:
Statement I:
 will undergo alkaline hydrolysis at a faster rate than
will undergo alkaline hydrolysis at a faster rate than 
Statement II:
In  intramolecular substitution takes place first by involving lone pair of electrons on nitrogen.
intramolecular substitution takes place first by involving lone pair of electrons on nitrogen.
Consider the following sequence of reactions to produce major product (A):

The molar mass of the product (A) is g mol−1. (Given molar mass in g mol−1 of C: 12,
H: 1, O: 16, Br: 80, N: 14, P: 31)
If 1 mM solution of ethylamine produces pH = 9, then the ionization constant ( ) of ethylamine is .
The value of x is (nearest integer).
The degree of ionization of ethylamine can be neglected with respect to unity.
Match the LIST-I with LIST-II

![product [A], [B], and [C] in the following reaction](https://images.collegedunia.com/public/qa/images/content/2025_03_17/Screenshot_029098311742200193386.jpeg)
![product [A], [B], and [C] in the following reaction](https://images.collegedunia.com/public/qa/images/content/2025_03_17/Screenshot_058819f71742200255416.jpeg)
![product [A], [B], and [C] in the following reaction](https://images.collegedunia.com/public/qa/images/content/2025_03_17/Screenshot_25adc5291742200411302.jpeg)
![product [A], [B], and [C] in the following reaction](https://images.collegedunia.com/public/qa/images/content/2025_03_17/Screenshot_ca6d85501742200449737.jpeg)
![product [A], [B], and [C] in the following reaction](https://images.collegedunia.com/public/qa/images/content/2025_03_17/Screenshot_5fcadf531742200485208.jpeg)
Choose the correct sets with respective observations:
(1) (acidified with acetic acid) + (neutralized with NaOH) → Blue precipitate
(2) + → Blue precipitate
(3) + →
(4) + → 6KC1
In the light of the above options, choose the correct set:
+ → 6KC1
Given below are two statements:
Statement (I): In octahedral complexes, when high spin complexes are formed. When low spin complexes are formed.
Statement (II): In tetrahedral complexes because of , low spin complexes are rarely formed.
In the light of the above statements, choose the most appropriate answer from the options given below:
Quantitative analysis of an organic compound (X) shows the following percentage composition.
C: 14.5%
Cl: 64.46%
H: 1.8%
Empirical formula mass of the compound (X) is:
The compounds that produce CO with aqueous NaHCO solution are: 
Both acetaldehyde and acetone (individually) undergo which of the following reactions?
A. Iodoform Reaction
B. Cannizzaro Reaction
C. Aldol Condensation
D. Tollen's Test
E. Clemmensen Reduction
Choose the correct answer from the options given below:
Given below are two statements:
Statement I: D-glucose pentaacetate reacts with 2,4-dinitrophenylhydrazine.
Statement II: Starch, on heating with concentrated sulfuric acid at 100°C and 2-3 atmosphere pressure, produces glucose.
In the light of the above statements, choose the correct answer from the options given below:
A molecule (P) on treatment with acid undergoes rearrangement and gives (Q). (Q) on ozonolysis followed by reflux under alkaline condition gives (R). The structure of (R) is given below. The structure of (P) is: 
The products A and B in the following reactions, respectively, are: 
ii)
iii)
The hydrocarbon (X) with molar mass 80 g mol and 90% carbon has degree of unsaturation.
The observed and normal molar masses of compound MX are 65.6 and 164 respectively. The percent degree of ionisation of MX is % (Nearest integer).
Identify correct statement/s:
(A) and are activating groups.
(B) and are meta directing groups.
(C) and are meta directing groups.
(D) Activating groups act as ortho- and para-directing groups.
(E) Halides are activating groups.
Choose the correct answer from the options given below:
Match List - I with List - II: 

0.1 mole of compound S will weigh ....... g,
(given the molar mass in g mol :
If is 30% ionised in an aqueous solution, then the value of van't Hoff factor is:
Match List - I with List - II:

Choose the correct answer from the options given below:





A. Resonating structures with more covalent bonds and less charge separation are more stable.
B. In electromeric effect, an unsaturated system shows +E effect with nucleophile and –E effect with electrophile.
C. Inductive effect is responsible for high melting point, boiling point and dipole moment of polar compounds.
D. The greater the number of alkyl groups attached to the doubly bonded carbon atoms, higher is the heat of hydrogenation.
E. Stability of carbanion increases with increase in s-character of the carbon carrying negative charge.

The percentage of nitrogen in the yellow product (X) formed is ________ %. (Nearest Integer)
(Given Molar mass in H:1, C:12, N:14)
$ \text{BaSO}_4 \text{BaSO}_4 x ^{-1} \text{BaSO}_4 \Lambda_m = \Lambda_m^\circ$)
$ K_c B(g) A(g) B(g) R = 0.082 \text{ L atm K}^{-1} \text{ mol}^{-1}$)


- Alcohol being less polar solvent as compared to water, alcoholic KOH favours elimination reaction with R–X.
- Order of reactivity towards mechanism is .
- Non-substituted aryl halides exhibit properties similar to alkyl halides.
- Vinyl chloride is an example of haloalkene and allyl chloride is an example of haloalkyne.
- can be prepared by reacting with but cannot be prepared by reacting with .
Match the column [
Statement-II: On nitration of aniline, the meta product is formed more than the ortho product.



Statement - II: , O-CH bond cleavage takes place during reaction with HI.

Where: (X) is the C=O bond, (Y) is the C-O bond between carbonyl group and oxygen, (Z) is the C-O bond between the oxygen and the methyl group.


Statement II: Rose essence from rose petals is separated by steam distillation because of its higher volatility and insolubility in .



(b) Both forms have difference in configuration at carbon.
(c) -form has more melting point than -form.
(d) Specific rotation of -form is and -form has .
(e) -form crystallises at and -form crystallises at .
Statement-II: 2,2,6,6-Tetraethylcyclohexanone will show tautomerism. Choose the correct option:
If the reaction goes to 60% yield of X, find the number of moles of ‘X’ formed.



(A) n- \hspace{1cm} (B) n-
(C) n- \hspace{1cm} (D)
(B) n-
(C) n-
(D)


(B) , 773 K
(C) Benzyl chloride/AlCl
Find the number of π-bonds in the final product.
Statement-II: Methane can’t be prepared by unsaturated hydrocarbon and by Wurtz reaction. Choose the correct option:

B. p-nitrophenol
C. Ethanol
D. p-methoxyphenol

(a) Cis-but-2-ene
(b) 2,3-Dimethylbut-2-ene
(c) 2-Methylbut-2-ene
(d) Propene
IUPAC name of major product will be: 
reacts with and forms which reacts with Na in the presence of dry ether to form . Find the molecular mass of .
Statement-I: Glucose exists in two anomeric.
Statement-II: In open chain structure at C-3, C-4, C-5, glucose and fructose have identical configuration. 
(A) If two orbitals are having the same value of , then the orbital having the lower value of will have lower energy.
(B) If atomic number increases, then the energy of orbitals belonging to a particular shell increases.
(C) Among 4s, 5d, 6f and 5p orbitals, none of these orbitals have two radial nodes.
(D) Size of 2 orbital is less than 3 orbital.
Which of the following statements are correct?
(a) Alkyl amide do not react.
(b) Secondary amide do not form secondary amide.
(c) Ratio of NaOH and Br₂ is 4 : 2.
(d) Na₂CO₃, NaBr and H₂O also formed along with amine.


Statement-2: In fractionating column, one which has higher boiling point value will be more in concentration above the fractionating column.

Boiling point order is in decreasing order due to decreasing Vanderwaal's forces.

P-dichlorobenzene has more melting point value due to its symmetric structure than O-dichlorobenzene while it has less boiling point than O-dichlorobenzene.

Benzene reacts with CH₃-C-Cl / AlCl₃ to give 'Q'.
P and Q react with each other in presence of NaOH / H₂O give product Z. Find number of π e⁻ in product Z.
The % increase in oxygen in steam volatile product with respect to phenol is ____ .
Benzene reacts with propyl chloride in presence of to give product . Mark correct statement(s) for the given reaction. 
(a) One of the intermediate is formed due to rearrangement.
(b) Major product is n-propylbenzene.
(c) Polysubstitution of substrate is also possible.
(d) Electron releasing group decreases rate of reaction.
Hydrocarbon (P) on reductive ozonolysis gives products which give positive iodoform test and on acidification produces the compound shown. Identify the structure of (P). 
Compound A is formed when compound B reacts with reagent (R). When compound C reacts with the same reagent (R), the product formed will be (S). Identify reagent (R) and product (S). 
Which amongst the following will give benzyl isocyanide as a major product? 
i & ii
ii & iii
i & iii
ii & iv
Statement 1: Stability of carbanion follows the order shown.
Statement 2: Allylic and benzylic carbanion stability is based on inductive effect and not resonance effect.


Consider the following reaction of benzene. the percentage of oxygen is _______ %. (Nearest integer) 
Consider the reaction given below:

.
Write the correct order of rate of reaction of following compounds with
P: -dimethylaniline
Q: -dimethyl-3-methylaniline
R: -dimethyl-2,6-dimethylaniline
Most preferred site for electrophilic substitution in above example?
(Note: The molecule contains a nitrogen-containing ring and a carbonyl-containing ring, labels are U, S, R, P).
Statement-II : Azeotropic mixture cannot be separated by using fractional distillation.
Statement-2 : Positive charge on ethyl will be unstable.


Which of the following is most acidic? 
| Column-I (Name of test) | Column-II (Pair of compounds) |
| (i) Neutral test | (P) Aldehyde vs Ketone pairs |
| (ii) Isocyanide test | (Q) Phenol vs Ethanol pairs |
| (iii) Ammonical silver nitrate test | (R) Primary amine vs Secondary amine pairs |
| (iv) test | (S) Carboxylic acid vs Phenol pairs |
(i) Vit. → (P) Ascorbic acid
(ii) Vit. → (Q) Riboflavin
(iii) Vit. → (R) Thiamine
(iv) Vit. C → (S) Pyridoxine
(i) with and
Products obtained are:


Statement I : Compound (X), shown below, dissolves in solution and has two chiral carbon atoms.

Statement II : Compound (Y), shown below, has two carbons with hybridization, one carbon with and one carbon with hybridization.

In the light of the above statements, choose the correct answer from the options given below :
A. Concentration of Fe²⁺ is increased.
B. Concentration of Fe³⁺ is decreased.
C. Concentration of Fe²⁺ is decreased.
D. Concentration of Fe³⁺ is increased.
E. Concentration of Cl⁻ is increased.
Choose the correct answer from the options given below :
Statement I: Sublimation is used for the separation and purification of compounds with low melting point.
Statement II: The boiling point of a liquid increases as the external pressure is reduced.
In the light of the above statements, choose the correct answer from the options given below :




Consider the following ions:
The stability of the ions is in the order:
Consider the following two reactions A and B: 
The numerical value of [molar mass of + molar mass of ] is ___.
Consider the following reaction sequence: 
Given: Compound (x) has percentage composition , and vapour density . Compound (y) develops a characteristic colour with neutral solution. Identify the {INCORRECT statement.}




The correct order of acidic strength of the major products formed in the given reactions is: 
A. Ionic radii of trivalent cations of group 13 elements decreases down the group.
B. Electronegativity of group 13 elements decreases down the group.
C. Among the group 13 elements, Boron has highest first ionisation enthalpy.
D. The trichloride and triiodide of group 13 elements are covalent in nature.
Choose the correct answer from the options given below :

Consider the following reactions giving major product. Identify the correct reaction. 

If gram of reacts with , then of gas is obtained at STP. Calculate .

CaCO3 + 2HCl → CaCl2 + H2O + CO2
If 90 g CaCO3 is added to 300 mL of HCl which contains 38.55% HCl by mass and has density 1.13 g mL-1,
then which of the following option is correct?
Given molar mass of H, Cl, Ca and O are 1, 35.5, 40 and 16 g mol-1 respectively.
Statement I: is more stable than as nine hyperconjugation interactions are possible in .
Statement II: is less stable than as only three hyperconjugation interactions are possible in .
In the light of the above statements, choose the correct answer...
Which of the following reaction is NOT correctly represented? 
Consider the following reactions: 
The oxidation states of Cu in Z and Q, respectively are:
Which one of the following graphs accurately represents the plot of partial pressure of CS₂ vs its mole fraction in a mixture of acetone and CS₂ at constant temperature?

A. Sodium hypochlorite reacts with KI to give KOI.
B. KOI is a reducing agent.
C. -unsaturated methylketone ( ) will give iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.



Statement I: C–Cl bond is stronger in than in .
Statement II: The given optically active molecule, on hydrolysis, gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below:
\bigskip
Given below are two statements:
Statement I:  Aniline can be synthesized from
Aniline can be synthesized from  using simpler reagents in the order
using simpler reagents in the order
i) Acidic KMnO4,
ii) Ammonia,
iii) Bromine and alkali
Statement II:  Aniline can be converted into
Aniline can be converted into  using reagents in the order
using reagents in the order
i) Bromine-H2O
ii) NaNO2/HCl (0 - 5 C)
(iii) H3PO2.
In the light of the above statements, choose the correct answer from the options given below:

Ph—CH—NH (with CH substituent)
CH —CH—NH (with CH substituent)
CH —CH —CH—CH (with NH substituent)
(Given molar mass in g mol : H : 1, C : 12, N : 14, O : 16)

If ‘n’ moles of AgCl is formed during the reaction of the complex with formula
CoCl2(en)2NH3
with excess AgNO3 solution, then the number of electrons present in the t2g orbital of the complex is ________.




[(A)] Propanal and propanone are functional isomers
[(B)] Ethoxyethane and methoxypropane are metamers
[(C)] But-2-ene shows optical isomerism
[(D)] But-1-ene and but-2-ene are functional isomers
[(E)] Pentane and 2,2-dimethylpropane are chain isomers The correct statements are:
\hspace*{1.3cm}
\hspace*{1.3cm}
\hspace*{1.3cm}
\hspace*{1.3cm}








Statement-II: R–N≡C on hydrolysis gives RCOOH.
List-I (1) Baeyer's reagent
(2) Cerric Ammonium nitrate
(3) Tollen's reagent
(4) Neutral FeCl
List-II (P) Violet colour
(Q) Red
(R) Silver mirror
(S) Pink colour disappears



(i) KOH + Br
(ii) CHCl , NaOH
X will be:




Which of the following is the correct structure for A, B, and C?


[(I)] Deactivating towards electrophilic substitution
[(II)] Activating towards electrophilic substitution
[(III)] Deactivating towards nucleophilic substitution
[(IV)] Activating towards nucleophilic substitution
Find the correct statements:
Select the correct statement regarding compounds P, Q, R, S.
Structures: P = Acetophenone, Q = Phenylacetaldehyde, R = Cinnamyl alcohol, S = Acetophenone. 
Statement-II : will rotate the plane polarised light after solvation.